AP Syllabus focus: ‘Substances with similar intermolecular interactions tend to be miscible or soluble in one another, linking solubility trends to particle-level attractions.’
Predicting whether substances mix is a major application of intermolecular forces. “Like dissolves like” connects molecular structure to solubility by comparing which attractions are present and whether new solute–solvent attractions can replace old ones.
Core idea: matching intermolecular attractions
Dissolving requires disrupting some solute–solute attractions and some solvent–solvent attractions, then forming new solute–solvent attractions. Mixing is favoured when the new attractions are comparable in type and strength to those that were broken.
Solubility: The extent to which a solute dissolves in a solvent to form a homogeneous solution at a given temperature.
“Like” refers to similarity in intermolecular forces (IMFs) (and therefore similar charge distribution and polarizability), not simply similar chemical formulas.
The key IMF categories used for “like dissolves like”
Nonpolar substances (roughly symmetric, no net dipole): primarily London dispersion forces (LDF).
Polar molecular substances (net dipole): dipole–dipole attractions (plus LDF).
Hydrogen-bonding substances (H bonded to N, O, or F and a lone-pair acceptor present): strong, directional hydrogen bonding (plus other IMFs).
Ionic substances: strong ion–ion attractions in the solute; dissolution requires strong ion–dipole interactions with the solvent.
Soluble vs miscible: language you must use correctly
Miscible: Two liquids that mix in all proportions to form a single phase.
A liquid solute in a liquid solvent is described as miscible or immiscible; a solid (or gas) in a liquid is described using solubility (soluble, slightly soluble, insoluble).
Applying “like dissolves like” to common cases
Nonpolar solutes in nonpolar solvents
Nonpolar solutes (e.g., many hydrocarbons) tend to dissolve in nonpolar solvents because:
Breaking LDF in the solute and solvent is “paid for” by forming new LDF of similar magnitude.
Similar polarizability and surface contact enable effective dispersion attractions.
Polar solutes in polar solvents
Polar solutes tend to dissolve in polar solvents because:
Dipole–dipole attractions and alignment between partial charges can form readily.
If hydrogen bonding is possible, matching H-bond donors/acceptors increases compatibility.
Hydrogen-bonding compatibility
Hydrogen bonding is most effective when the solute and solvent can both participate.
Molecules with O–H or N–H groups often dissolve well in solvents that can accept H-bonds (lone pairs).
Molecules with only H-bond acceptors (e.g., a carbonyl oxygen) may dissolve better in solvents that can donate H-bonds.
Ionic solutes in polar solvents
Ionic compounds dissolve best in solvents with strong molecular dipoles (often water) because ion–dipole attractions can compensate for the energy needed to separate ions.
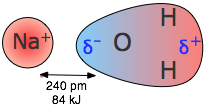
Ion–dipole attraction is shown by water molecules orienting their partial charges toward an ion: the oxygen end () points toward a cation like Na, while the hydrogen ends () point toward an anion. This directional alignment illustrates how hydration shells stabilize separated ions in solution, helping offset the energetic cost of disrupting an ionic lattice. Source
If ion–dipole attractions are too weak, the crystal lattice remains intact and solubility is low.
Solvation: The process in which solvent particles surround and interact with solute particles.
Hydration: Solvation specifically by water.
What to look for when predicting solubility from structure
Compare the “interaction inventory”
Ask which attractive forces are available for each substance:
Does the solute have a net dipole?
Can it donate an H-bond (N–H, O–H, F–H)?
Can it accept an H-bond (lone pairs on N, O, F)?
Is it largely nonpolar (hydrocarbon-like) with only LDF?
Does it form ions (ionic compound) requiring ion–dipole stabilization?
Balance of polar and nonpolar regions
Many molecules contain both polar and nonpolar parts. Solubility depends on which parts dominate the intermolecular behaviour:
Increasing nonpolar surface area often decreases solubility in very polar solvents.
Adding polar functional groups generally increases solubility in polar solvents by adding stronger solute–solvent attractions.
Why “like dissolves like” works (a thermodynamic lens)
Mixing is favoured when it leads to a negative Gibbs free energy change.
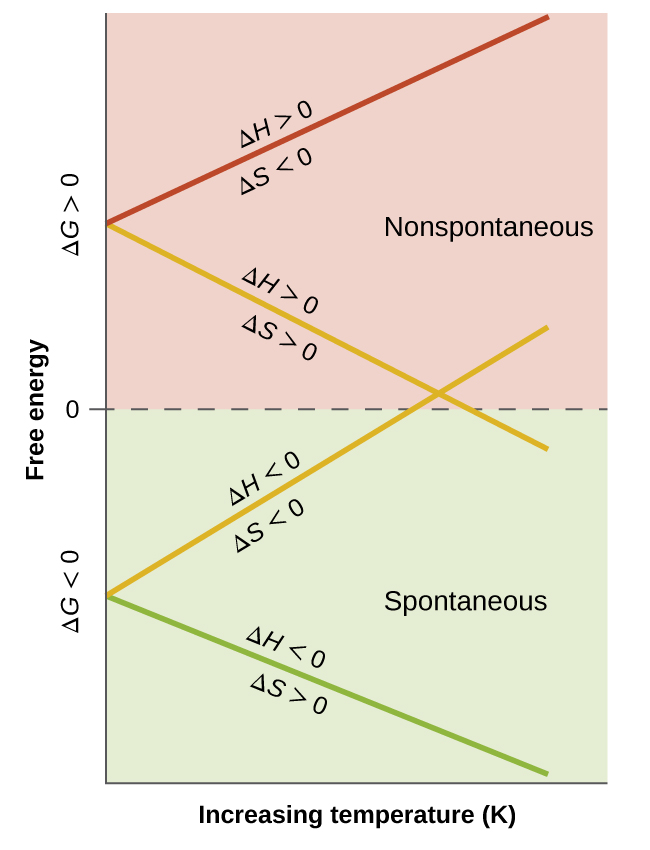
A vs. plot makes the equation visual: the intercept corresponds to and the slope corresponds to . The temperature where the line crosses marks the threshold between nonspontaneous and spontaneous behavior, clarifying why entropy can dominate at higher when . Source
= Gibbs free energy change for mixing (J mol)
= Enthalpy change from breaking/forming attractions (J mol)
= Temperature (K)
= Entropy change from dispersing particles (J mol K)
When IMFs “match,” is often small (or favourable), and dispersal tends to make positive, so dissolution is more likely.
Common pitfalls and boundaries of the rule
All molecules have LDF; the question is whether stronger, more specific attractions (dipole–dipole, H-bonding, ion–dipole) are needed to dissolve.
“Polar” is not all-or-nothing; small structural changes can shift the dominant interactions and change solubility noticeably.
Some mixtures form separate layers because solute–solvent attractions are too weak compared with solute–solute and solvent–solvent attractions, producing immiscibility even though each pure liquid is held together well.
FAQ
A higher dielectric constant better stabilises separated charges.
This strengthens ion–dipole stabilisation and often increases ionic solubility.
A co-solvent can introduce missing interactions (e.g., H-bond donation/acceptance) and “bridge” between polar and non-polar regions.
It can also change solvent structure around the solute.
Added ions strongly attract water molecules, reducing water’s ability to solvate non-polar solutes.
Non-polar solutes then aggregate because solute–solvent attractions become relatively weaker.
They contain both polar and non-polar regions.
They can form structures (e.g., micelles) where polar parts interact with water and non-polar parts cluster together.
If the solute forms very strong solute–solute attractions (e.g., extensive H-bond networks), water may not compensate enough by forming equally strong solute–solvent interactions.
Molecular size and shape can also limit effective solvation.
Practice Questions
(2 marks) Explain, in terms of intermolecular forces, why a non-polar solute is more likely to dissolve in a non-polar solvent than in water.
Mentions both solute and solvent are dominated by London dispersion forces, so new solute–solvent attractions are similar in type/strength. (1)
States water is polar and strongly attracted to itself (dipole/H-bonding), so replacing these with weak solute–water attractions is unfavourable. (1)
(5 marks) A student compares the solubility of two molecular solutes in water: Solute A has an –OH group and a short hydrocarbon chain; Solute B has only a longer hydrocarbon chain. Using “like dissolves like,” predict which is more soluble and justify using particle-level attractions.
Correct prediction: Solute A is more soluble in water. (1)
Identifies water as polar and capable of hydrogen bonding. (1)
Explains Solute A can form hydrogen bonds (donor/acceptor interactions) with water, strengthening solute–solvent attractions. (1)
Notes Solute B interacts mainly via London dispersion forces, which are weak compared with water–water attractions. (1)
States increasing non-polar surface area reduces compatibility with water, so longer hydrocarbon chains lower solubility. (1)

