AP Syllabus focus: ‘Dissolution involves reorganizing solvent around dissolved species, contributing to both ΔH° and ΔS° of the process.’
Dissolving a substance is not only “mixing.” Solvent molecules must rearrange around the incoming particles, changing intermolecular interactions and molecular freedom. This solvent reorganisation strongly influences both the enthalpy and entropy of dissolution.
What “solvent reorganisation” means
When solute particles separate into a solvent, nearby solvent molecules reorient, shift positions, and sometimes form structured layers. This rearrangement is necessary to stabilise the dissolved species and to maintain favourable solvent–solvent interactions elsewhere.
Solvation shell (hydration shell in water): The layer(s) of solvent molecules arranged around a dissolved particle due to solute–solvent attractions and solvent restructuring.

A cation’s electric field aligns a nearby dipolar molecule so its negative end points toward the ion and its positive end points away. This illustrates the microscopic origin of strong ion–dipole attractions and why solvent molecules in the first solvation shell adopt restricted orientations. Source
Solvent reorganisation happens for ions, polar molecules, and nonpolar molecules, but the dominant forces (and thermodynamic consequences) differ.
Why solvent reorganisation changes ΔH°
Enthalpy reflects the net energy change from breaking and making interactions.
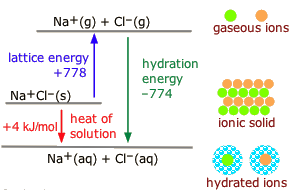
A thermodynamic cycle for dissolving an ionic solid in water partitions the overall enthalpy change into lattice separation and hydration (solute–solvent) steps. The diagram emphasizes that for dissolution is a balance between energy required to separate ions and energy released when water reorganizes to stabilize them. Source
Solvent reorganisation typically involves:
Disrupting solvent–solvent attractions (e.g., hydrogen bonding in water) to create space for solute particles
Orienting solvent dipoles around solute particles (especially ions), which can be energetically favourable
Straining or strengthening the remaining solvent network as it adjusts
Key qualitative ideas:
Strong ion–dipole attractions can make solvent reorganisation enthalpically favourable (more negative contribution to ).
If creating a cavity requires breaking many strong solvent–solvent interactions, solvent reorganisation can be enthalpically unfavourable (positive contribution to ).
Highly charged, small ions (high charge density) tend to cause larger solvent rearrangements and stronger local ordering, often with a larger enthalpy change.
Why solvent reorganisation changes ΔS°
Entropy tracks how dispersed matter/energy is and how many molecular arrangements are available. Solvent reorganisation affects entropy by changing solvent “freedom”:
Solvent molecules in a solvation shell may have restricted orientations (fewer microstates)
Surrounding solvent may become more or less structured depending on solute type
Mixing itself tends to increase dispersal, but local ordering can offset that
General trends students should be able to argue:
Ionic solutes in water: hydration shells often impose order on nearby water molecules, giving a negative (unfavourable) contribution to .
Nonpolar solutes in water: water may form more ordered arrangements around nonpolar surfaces (often described as increased structuring), which can also reduce .
If dissolution disrupts an existing solvent structure substantially and increases overall freedom of solvent molecules, solvent reorganisation can contribute positively to .
Connecting reorganising solvent to overall favourability
Solvent reorganisation is one part of the overall thermodynamic balance for dissolution because it simultaneously influences enthalpy and entropy.
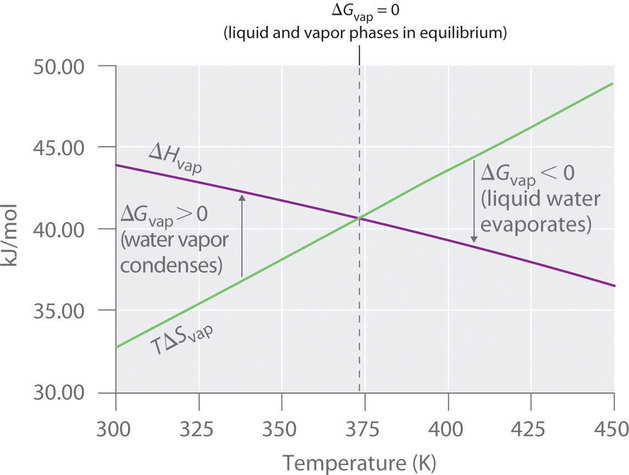
A temperature-dependent plot comparing and shows how their competition controls the sign of . The crossover point (where ) marks , reinforcing that spontaneity depends on the balance of enthalpic and entropic terms. Source
= standard Gibbs free energy change, kJ/mol
= standard enthalpy change, kJ/mol
= temperature, K
= standard entropy change, kJ/(mol·K)
A common qualitative outcome is compensation: solvent reorganisation may be enthalpically favourable (strong attractions) but entropically unfavourable (more ordering), or vice versa.
How to reason qualitatively on AP-style prompts
Use particle-level statements tied to thermodynamics:
Identify whether solvent molecules must strongly orient around the solute (suggests decreased entropy).
Compare the strength of new solute–solvent attractions to the solvent–solvent attractions that must be disrupted (suggests the sign of the enthalpy contribution).
Mention that these effects are local (near the solute) but can be large enough to control observed solubility and temperature dependence.
FAQ
Mainly ion charge density and solvent polarity.
Smaller, more highly charged ions typically organise more layers of water than larger, singly charged ions.
Yes.
Polar solutes align nearby solvent dipoles; nonpolar solutes can cause surrounding water to adopt more structured arrangements compared with bulk water.
Mixing increases dispersal, but local solvent ordering near solute particles reduces the number of accessible orientations.
The net $\Delta S^\circ$ reflects the balance of these effects.
Because the entropy term is multiplied by $T$.
If solvent reorganisation makes $\Delta S^\circ$ negative, increasing $T$ makes $-T\Delta S^\circ$ more positive, which can reduce favourability.
Large temperature dependence of solubility and noticeable heat absorption/release on dissolving can indicate significant restructuring.
Changes in viscosity or hydration-number measurements (from spectroscopy) also reflect strong solvation shells.
Practice Questions
Explain why forming hydration shells around ions can make for dissolution less positive (or more negative).
States that water molecules become more ordered/oriented around ions in hydration shells (1)
Links this ordering to fewer arrangements/microstates, so entropy decreases (1)
A salt dissolves in water. Describe how solvent reorganisation can contribute to both and for the dissolution, referring to interactions and molecular motion.
Breaking/disrupting some water–water (hydrogen-bond) interactions to make space affects (1)
Forming ion–dipole attractions between ions and water affects (1)
Hydration shells restrict water orientation/rotation near ions, decreasing entropy (1)
Notes entropy/enthalpy “compensation” is possible (one favourable, one unfavourable) (1)
Uses clear particle-level language (reorientation/restructuring of solvent) tied to thermodynamic terms (1)

