AP Syllabus focus: ‘Use tabulated standard molar entropies (S°) for each species to describe how dispersed energy and matter are under standard conditions.’
Entropy tables let you compare how energy and particles are distributed in different substances. In AP Chemistry, you interpret standard molar entropy values under standard conditions to reason about relative dispersal.
What “absolute entropy” means
Entropy as a state function
Entropy (S): a thermodynamic state function that quantifies how dispersed energy and matter are among the possible microscopic arrangements (microstates) of a system.
Because entropy is a state function, each substance at a specified temperature, pressure, and physical state can be assigned a definite entropy value.
Absolute (standard molar) entropy,
Standard molar entropy (): the entropy of 1 mole of a substance in its standard state at a stated temperature (commonly 298 K), reported in J·mol⁻¹·K⁻¹.
A key AP idea is that values are tabulated (like ), so you can look them up to compare how dispersed energy and matter are for each species under the same reference conditions.
Standard conditions and standard states (AP Chemistry usage)
What “standard” refers to for entropy tables
When an entropy table lists , it assumes each species is in its standard state at the temperature specified by the table (often 298.15 K). In AP Chemistry, standard state conventions are:
Gases: pure gas at 1 bar (or often approximated as 1 atm in coursework)
Solutions: 1.0 M (for species whose standard state is defined in solution)
Pure liquids and pure solids: the pure substance at 1 bar
Elements: their most stable form at 1 bar and the stated temperature (e.g., C(graphite), not C(diamond))
In this subtopic, the essential skill is using the tabulated for each species to describe relative dispersal under those standard-state assumptions.
How to interpret tabulated values
Units and meaning
is reported per mole per kelvin: J·mol⁻¹·K⁻¹
Larger means, under standard conditions, the substance has:
more accessible microstates (more ways to distribute energy)
greater particle/energy dispersal
Reliable comparisons you can make
Using tables, you can compare species at the same temperature (e.g., 298 K) and infer which has higher entropy.
Common qualitative patterns (used for interpreting tables, not for calculating):
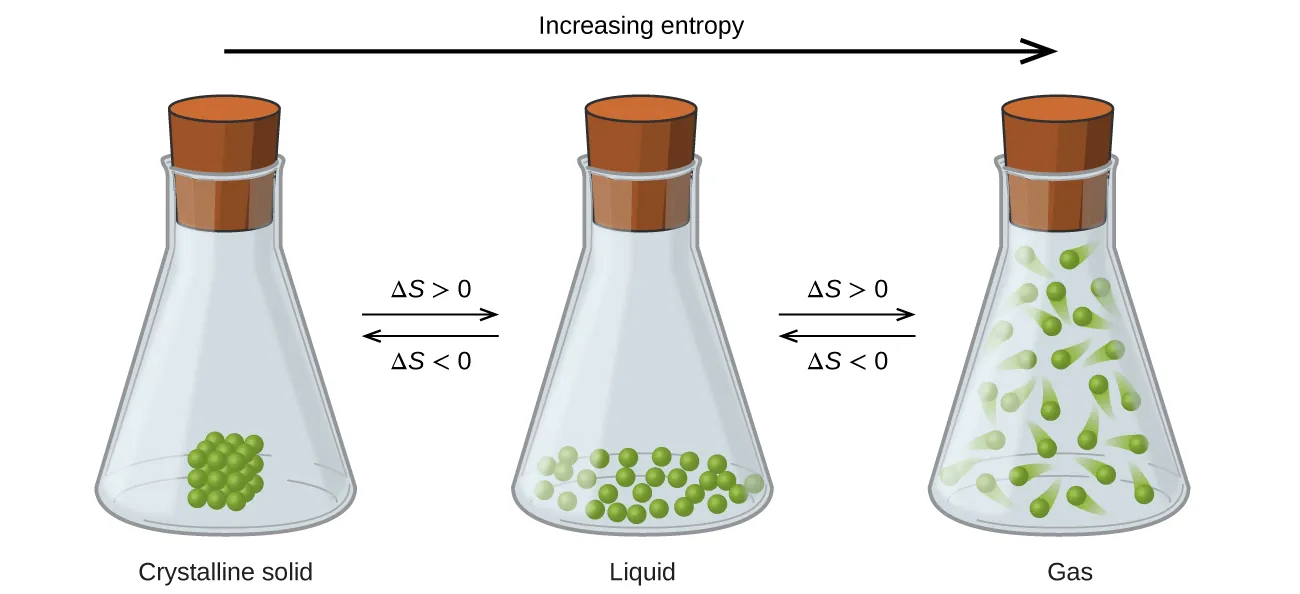
Phase changes strongly affect dispersal because each phase offers a different number of accessible microstates. The diagram shows that entropy increases as a substance goes from a crystalline solid to a liquid to a gas, consistent with for the same substance at the same . Source
Physical state (same substance, same T): typically (s) < (l) < (g) because gases have far more possible arrangements and energy distributions.
Molar mass/complexity (same phase): larger, more complex molecules often have larger because they have more motions (rotations/vibrations) and configurations.
Allotropes/structures: a more “rigid” or more ordered crystal structure can have a smaller than a less ordered one, even for the same element.
Important cautions
Do not confuse (absolute entropy) with (entropy change); tables give for individual species.
values depend on temperature; always check the table’s temperature (AP problems most often use 298 K).
For entropy, unlike enthalpy of formation, the standard value for an element in its standard state is not zero at 298 K; elements have substantial dispersal at ordinary temperatures.
Why values are not zero at room temperature
The reference point for “absolute” entropy is tied to the Third Law of Thermodynamics: a perfect crystal at 0 K has zero entropy.
A temperature–entropy (–) diagram illustrates how absolute entropy can be anchored by the Third Law, which sets the reference at for a perfect crystal at . The curve emphasizes that as temperature increases from 0 K, entropy rises because more energetic and positional microstates become accessible, making room-temperature values positive and measurable. Source
At 298 K, essentially all substances have thermal motion, so their values are positive and measurable—hence the usefulness of tabulated for describing dispersal under standard conditions.
FAQ
$S^\circ$ is an absolute property referenced to 0 K (Third Law), not a relative “formation” scale. At 298 K, elements still have thermal motion and many accessible microstates, so $S^\circ > 0$.
It is obtained from temperature-dependent heat capacity data. Conceptually, chemists determine how much reversible heat is absorbed as temperature increases, then integrate the heat-capacity behaviour from near 0 K to the target temperature.
Crystal structure and molecular flexibility matter. Rigid, highly ordered lattices reduce accessible microstates, while flexible molecules with many conformations increase microstates and raise $S^\circ$.
Yes, slightly. Gas entropies depend on pressure, so a different standard pressure shifts tabulated values by a small amount; AP Chemistry typically treats 1 atm and 1 bar as effectively equivalent unless stated.
Entropy increases with temperature, and tables are temperature-specific. Using a 298 K table at another temperature mixes reference states and can lead to incorrect comparisons of dispersal.
Practice Questions
(2 marks) Under standard conditions at 298 K, which has the larger standard molar entropy, : or ? Briefly justify your choice.
has larger (1)
Justification: ions are more dispersed/more possible arrangements in solution than in an ordered crystal lattice (1)
(5 marks) A student compares tabulated values at 298 K for , , and . Explain, using entropy as dispersal of energy and matter under standard conditions, why these substances can have very different values. Include at least three distinct points.
Correct link to meaning of as dispersal/microstate availability under standard conditions (1)
Phase effect: gases generally have higher than solids due to much greater freedom/dispersion (1)
Molecular complexity: more atoms/more vibrational and rotational modes increase (1)
Comparison among gases: typically has higher than due to greater molar mass and additional rotational/vibrational degrees of freedom (1)
Clarifies that values are tabulated for each species in its standard state at the stated temperature (1)

