AP Syllabus focus: ‘E° corresponds to Q = 1. As the system approaches equilibrium, |E| decreases and reaches zero at equilibrium when Q = K.’
Electrochemical cell potentials depend on how far a redox system is from equilibrium. This page connects reaction quotient (Q), equilibrium constant (K), and the condition where the measured cell potential E becomes zero.
Core ideas: Q, K, and what E is telling you
Interpreting cell potential under nonstandard conditions
A cell’s measured voltage E reflects the thermodynamic “push” for the overall redox reaction to proceed as written.
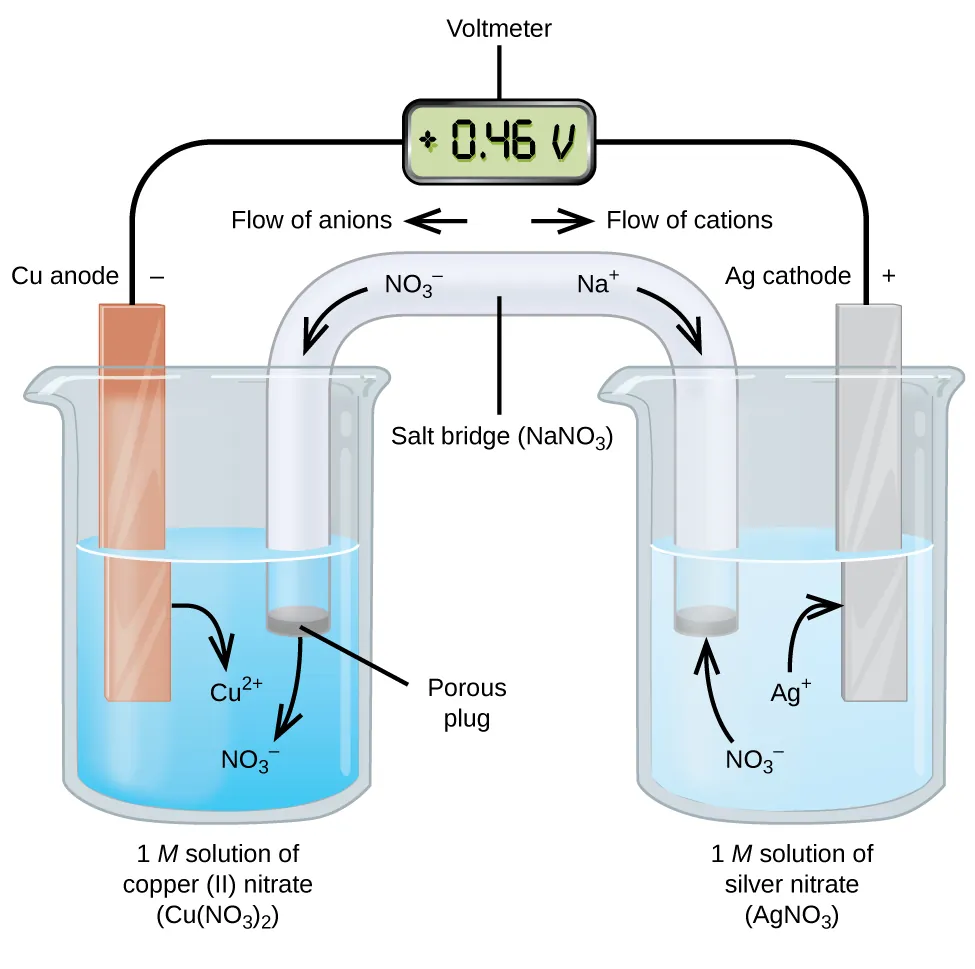
A standard galvanic cell schematic showing the anode/cathode, electron flow through the external circuit, and counter-ion migration through the salt bridge to maintain electroneutrality. This makes it clear what the measured cell potential represents experimentally and why changing solution composition (and thus ) is inseparable from the cell’s operation. Source
As the mixture composition changes during operation, the driving force changes too.
When the system is far from equilibrium, the driving force is larger and the magnitude of E is larger.
As products build up (or reactants are depleted), the driving force decreases and E moves toward zero.
At equilibrium, there is no net driving force for reaction progress, so E is zero.
Q (reaction quotient) describes the current composition
Reaction quotient (Q): The ratio of activities (often approximated by concentrations for solutes and partial pressures for gases) of products to reactants, each raised to their stoichiometric coefficients, at the current (not necessarily equilibrium) conditions.
Q changes continuously as the cell runs because reactant and product amounts change.
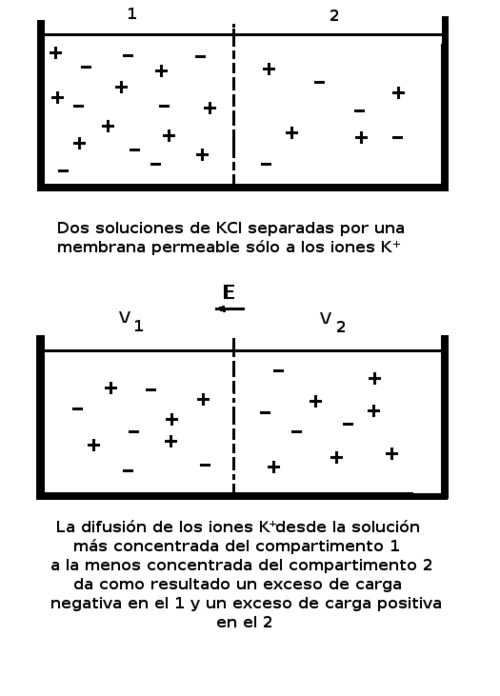
A schematic illustrating how a concentration (activity) difference across a semipermeable barrier generates a Nernst potential. The key takeaway is that electrochemical potential depends logarithmically on a ratio of activities/concentrations—an idea that directly parallels how depends on in the Nernst equation for cells. Source
K (equilibrium constant) is the special value of Q at equilibrium
Equilibrium constant (K): The value of Q when the reaction mixture has reached equilibrium at a given temperature (a temperature-dependent constant for a specified balanced reaction).
K is fixed for a given reaction at a fixed temperature, while Q depends on the moment-to-moment composition.
Standard conditions and why E° matches Q = 1
What “standard cell potential” corresponds to
The standard cell potential E° is defined for standard-state conditions of the participating species (so the composition is set to the standard reference choices). Under these conditions, the way Q is constructed makes it equal to 1 for the overall cell reaction.
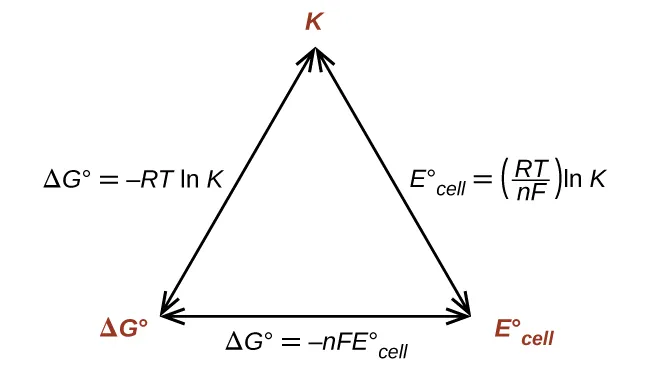
A relationship map connecting , , and the standard cell potential , emphasizing that any one of these quantities can be used to compute the other two. This helps students see that is not “just a voltage,” but a thermodynamic measure that is directly tied to equilibrium (via ) under standard conditions. Source
E° corresponds to Q = 1 because the standard-state activities are defined as 1 (so the product of activities in the Q expression becomes 1).
E° is therefore the cell’s potential when the reaction mixture is at the standard reference composition, not when it is at equilibrium (unless K also happens to be 1).
Approaching equilibrium: why |E| decreases and E reaches zero
The key trend
As the system approaches equilibrium, Q moves toward K. The cell potential responds by shrinking in magnitude.
If the cell reaction is initially product-favored at the starting conditions, reaction progress increases Q.
As Q gets closer to K, the net driving force decreases.
When Q finally equals K, the system is at equilibrium and E is zero.
This statement applies to the overall cell reaction written in the direction you are considering; reversing the reaction inverts Q and replaces K with 1/K.
Mathematical connection (what you should recognise)
= cell potential (V)
= standard cell potential (V)
= gas constant ()
= temperature (K)
= moles of electrons transferred in the balanced overall reaction (mol e)
= Faraday constant ()
= reaction quotient (unitless, based on activities)
= natural logarithm
= equilibrium constant (unitless)
In words: increasing Q (more products relative to reactants for the reaction as written) makes the subtraction term larger, so E decreases. At equilibrium, the formula predicts E = 0 exactly when Q = K, matching the syllabus statement.
Common AP-level reasoning checkpoints
Using Q, K, and the sign/magnitude of E
Compare Q to K to predict the direction the system must move to reach equilibrium.
The closer Q is to K, the closer E is to zero (smaller magnitude, weaker driving force).
At equilibrium, E is zero, but that does not mean concentrations are equal—only that Q has the equilibrium value K.
FAQ
$E^\circ$ reflects the standard-state composition ($Q=1$), not the equilibrium composition.
As the cell runs, composition changes, so $Q$ changes until $Q=K$, where $E=0$.
The condition $E=0$ still occurs at $Q=K$, but $K$ itself changes with temperature.
So the equilibrium composition (and therefore the required $Q$ value) shifts when $T$ changes.
In rigorous thermodynamics, pure solids and pure liquids have activity 1.
They therefore do not appear in $Q$, so changing their amounts does not shift $Q$ or the $Q=K$ condition.
The system is near equilibrium: $Q$ is close to $K$.
There is still a net driving force, but it is weak, so only a small change in composition is needed to reach equilibrium.
Not always; the sign depends on how the overall reaction is written and on $E^\circ$.
However, for a fixed reaction direction, moving $Q$ towards $K$ drives $E$ towards 0, and the magnitude typically shrinks as $Q$ approaches $K$.
Practice Questions
For a galvanic cell reaction at a fixed temperature, state the values of and at equilibrium. (Do not calculate.)
at equilibrium (1)
at equilibrium (1)
A student claims: “Because is measured under standard conditions, the cell must be at equilibrium when .” Using , , and the condition for , assess whether the claim is correct.
Recognises corresponds to under standard-state conditions (1)
States equilibrium condition is (1)
States at equilibrium (1)
Explains does not generally mean equilibrium unless (1)
Links “approaches equilibrium” to decreasing towards zero as (1)

