AP Syllabus focus: ‘“Hydrogen ion” and “hydronium ion” are used interchangeably in water; H3O+ is preferred, but H+(aq) is also accepted on the AP Exam.’
Chemists often write acids in water using either H3O+ or H+(aq). Understanding what each symbol really means helps you interpret acid–base equations, communicate clearly, and avoid common conceptual errors on AP Chemistry.
What the two notations mean in water
In aqueous solution, a “bare” proton does not exist as an isolated particle for any meaningful length of time. Instead, it is associated with water molecules.
Hydronium ion (H3O+): The aqueous species formed when a proton is bonded to a water molecule; it represents the acidic form of water in solution.
The AP Chemistry convention is to treat H3O+ as the more chemically explicit notation, while still allowing H+(aq) as a convenient shorthand.

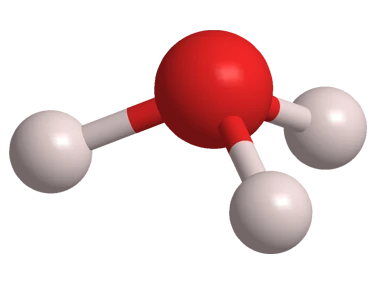
Ball‑and‑stick model of the hydronium ion, , showing oxygen at the center bonded to three hydrogens in a trigonal‑pyramidal arrangement. This visual reinforces that the “acidic proton” in water is represented as protonated water rather than a free, isolated . Source
Why H+(aq) is still written (and accepted)
You will frequently see H+(aq) in textbooks and reaction equations because it is compact and often makes stoichiometry clearer. On the AP Exam, H+(aq) is acceptable specifically because it is understood to mean “the proton as it exists in water,” not a free proton in a vacuum.
Key idea for AP Chemistry writing:
Preferred in water: H3O+(aq)
Also accepted: H+(aq) (interpreted as an aqueous proton, effectively hydronium)
Connecting the notations with a chemical statement
A useful way to connect the two is to show that “adding H+ to water” produces hydronium.
= aqueous proton shorthand (represents a proton solvated by water; no unit)
= liquid water solvent (no unit)
= hydronium ion, the explicit acidic species in water (no unit)
This relationship explains why the syllabus states that “hydrogen ion” and “hydronium ion” are used interchangeably in water.
How to write acid–base reactions consistently
When writing reactions in aqueous solution, either notation can be used as long as you are consistent and chemically sensible.
Common acceptable choices
For net ionic neutralisation, you may see either:
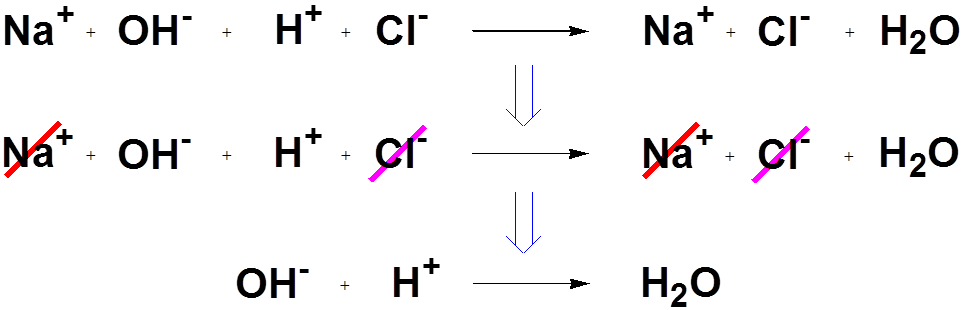
Diagram showing the cancellation of spectator ions to obtain the net ionic equation for strong acid–strong base neutralization. The figure emphasizes that the key chemical change is , matching the common AP Chemistry bookkeeping approach for aqueous acids and bases. Source
H+(aq) + OH−(aq) → H2O(l)
H3O+(aq) + OH−(aq) → 2 H2O(l)
When emphasising “acid in water,” H3O+ highlights that water is the proton acceptor.
Consistency tips
If you write H3O+ on the reactant side, ensure products reflect water correctly (often forming H2O).
If you write H+(aq), remember it is a bookkeeping shorthand for acidity in water, not a standalone particle.
Common pitfalls to avoid
Treating H+(aq) as a literal free proton rather than a water-associated species.
Mixing H+ and H3O+ within the same equation without adjusting water molecules, which can break atom balance.
Forgetting that the syllabus preference is H3O+, even though H+(aq) is accepted on the AP Exam.
FAQ
Not exactly. The proton is better pictured as dynamically solvated (e.g., clusters like $H_5O_2^+$ and $H_9O_4^+$). $H_3O^+$ is the simplest representative.
Choose one style and balance accordingly. If using $H_3O^+$, you will often need explicit $H_2O$ molecules to conserve H and O atoms.
Context. In aqueous acid–base chemistry it is usually implied to be aqueous, but adding “(aq)” reduces ambiguity, especially when comparing phases or reaction conditions.
It can change how you write them, but not the underlying chemistry. In water, $[H^+]$ is commonly treated as shorthand for the hydronium-related acidity.
No. $H_3O^+$ specifically requires water. In other solvents, the proton associates with that solvent instead, so a different conjugate acid species would be more appropriate.
Practice Questions
(2 marks) In aqueous solution, why is often written instead of , and which notation(s) are accepted on the AP Chemistry exam?
1 mark: States that in water the proton is associated with water as (no free proton).
1 mark: States is preferred but is also accepted.
(5 marks) A student writes an aqueous acid reaction using throughout.
(a) Explain what represents in water. (2 marks)
(b) Write an equation showing how this species relates to . (1 mark)
(c) Give two reasons a chemist might still choose to use in written equations. (2 marks)
(a) 1 mark: Identifies as an aqueous proton (hydrated/solvated), not a free proton.
(a) 1 mark: Links it conceptually to hydronium/protonated water.
(b) 1 mark: (or equilibrium form).
(c) 1 mark each: Any two of: simpler stoichiometry/bookkeeping; compact notation; common convention in net ionic equations; avoids explicitly writing solvent water repeatedly.

