AP Syllabus focus: ‘Le Châtelier’s principle can be used to predict how stresses affect experimentally measurable properties, including pH, temperature, and solution color.’
Chemical equilibria respond to disturbances by shifting in the direction that reduces the imposed stress. Those shifts are not just abstract—they can be tracked through real measurements such as pH, temperature, and solution color.
Connecting equilibrium shifts to what you can measure
What a “shift” means experimentally
A shift describes a net change in composition as the system moves toward a new equilibrium after a stress. Even though both forward and reverse reactions continue, measurable properties change because concentrations of key species change.
Common measurement signals include:
pH changes when changes
Temperature changes when heat is absorbed or released during re-equilibration
Color changes when the relative amounts of differently colored species change
pH as an indicator of equilibrium shifts
Using pH to track acid–base equilibria
In equilibria involving acids/bases, a stress that changes or shifts the equilibrium and produces a measurable pH change.
pH: A logarithmic measure of hydronium ion concentration; lower pH means higher .
pH is especially useful because small composition shifts can produce noticeable pH changes.
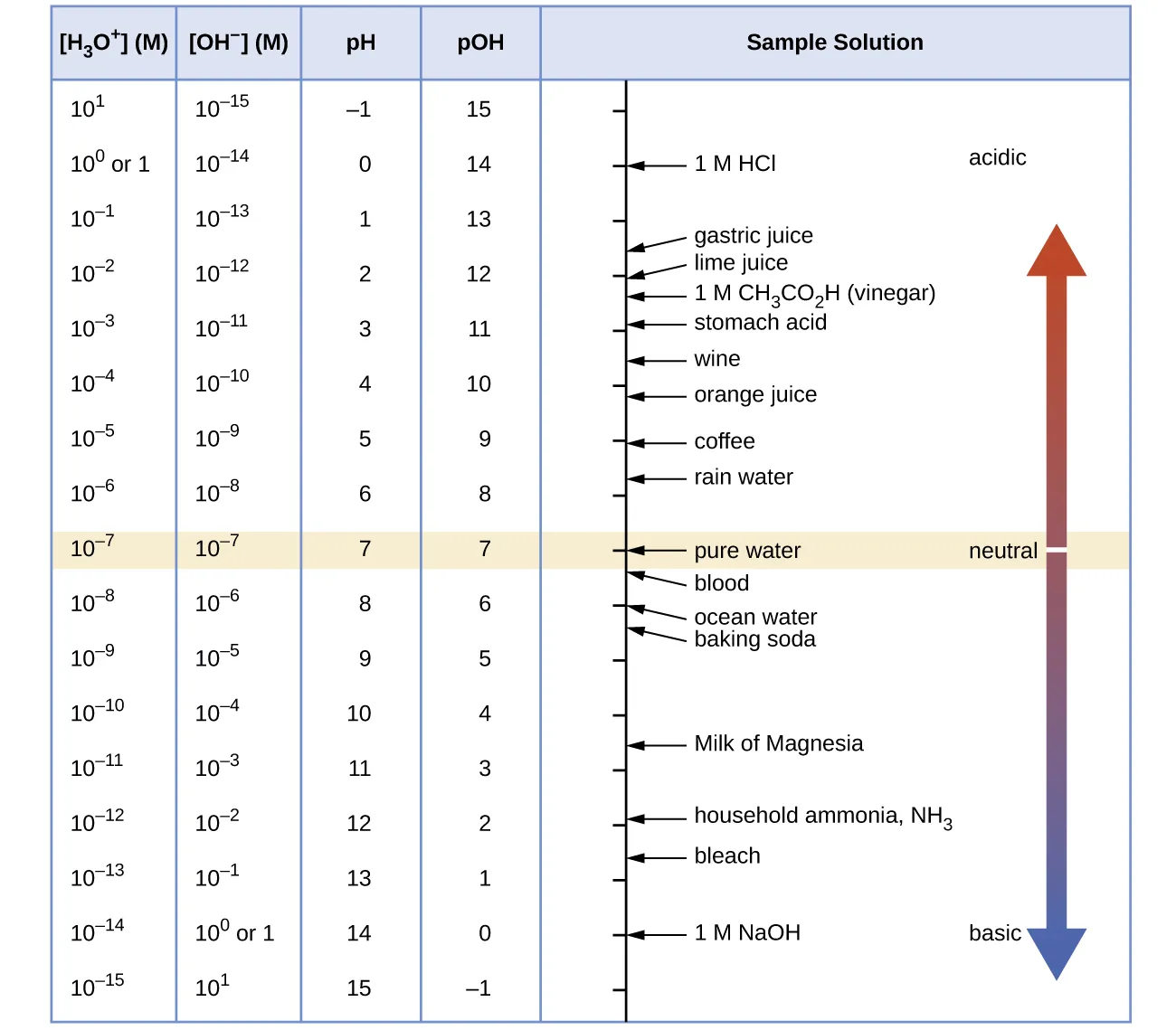
pH and pOH scales vs ion concentration (OpenStax, Fig. 14.2). The chart explicitly links and (in molarity) to their corresponding pH/pOH values on a logarithmic scale. It also anchors the scale with familiar reference substances, helping you interpret how concentration changes map to measurable pH changes. Source
= hydronium ion concentration (mol L)
Practical interpretation of pH-based shifts:
If a stress increases , pH decreases, indicating a shift that produces more acid (or consumes base).
If a stress decreases , pH increases, indicating a shift that consumes acid (or produces base).
Buffers show smaller pH changes because equilibrium shifts partially counter the stress; the remaining change is still measurable.
Temperature as an indicator of equilibrium shifts
Heat effects you can observe
When a stressed system re-establishes equilibrium, it may absorb or release heat depending on which direction is favoured for reducing the stress. A thermometer (or simply the container feeling warmer/cooler) can reveal the direction of net change.
How to connect temperature measurements to shifts:
If the system warms during re-equilibration, the net shift is producing heat (a heat-releasing direction).
If the system cools, the net shift is consuming heat (a heat-absorbing direction).
In well-insulated (nearly adiabatic) setups, temperature changes are more noticeable; in open setups, heat exchange with surroundings can mask the signal.
Solution color as an indicator of equilibrium shifts
Linking colour intensity and hue to composition
Many equilibria involve species with different colours (commonly aqueous complexes).
A stress that changes the relative concentration of these species leads to a visible color change as equilibrium is re-established.
Ways colour can change measurably:
Hue change: dominance shifts from one coloured species to another (e.g., yellow species increases while blue decreases).
Intensity change: one coloured species becomes more concentrated, deepening the colour.
Spectrophotometry (conceptually): absorbance changes reflect changing concentration of the coloured species, consistent with a shift.
How to reason from colour observations:
Identify which side contains the more strongly coloured species.
A deeper appearance of that colour indicates a shift toward that species; fading indicates a shift away.
FAQ
Colour intensity depends on both concentration and path length.
In practice, compare under identical conditions (same container, lighting, volume), or use absorbance to separate “looks darker” from “is more concentrated”.
Because pH is logarithmic: a modest change in $[H_3O^+]$ can shift pH noticeably.
Systems with low buffering capacity show larger pH swings for the same disturbance.
Heat exchange with the surroundings can overwhelm small heat effects.
Stirring, delayed readings, and warm glassware can also blur or reverse the apparent direction of temperature change.
Yes. Apparent colour can change due to dilution (lower overall concentration), different lighting, or precipitation/cloudiness scattering light.
A true equilibrium-composition change typically shows consistent, reversible trends upon repeating the stress.
Absorbance provides a quantitative signal that correlates with concentration of a coloured species.
It reduces subjectivity and can detect small shifts that are hard to see by eye, especially when colours overlap.
Practice Questions
A system at equilibrium contains an acid–base conjugate pair and the pH is monitored. After a small disturbance, the pH decreases. Using Le Châtelier’s principle, state what must have happened to and what this implies about the net direction of shift.
States increased (1)
Links increased to decreased pH (1)
States equilibrium shifted in the direction that produces / increases acidity (1)
An equilibrium mixture involves two aqueous species, one blue and one yellow. After a stress is applied, the solution becomes greener and the temperature of the mixture drops slightly. Explain, using Le Châtelier’s principle, how these observations can be used to infer the direction of the shift and the heat effect of the net change.
Explains green indicates changed ratio of blue and yellow species (1)
Infers a shift toward the species whose contribution to green increased (e.g., towards yellow if mixture looks more yellow-green; accept either with consistent reasoning) (1)
Links colour change to a composition change during re-establishing equilibrium (1)
Interprets temperature drop as net heat absorption during re-equilibration (1)
States the shift proceeds in the direction that reduces the imposed stress and is consistent with absorbing heat (1)
Uses correct chemical language (equilibrium, shift, stress, net change) (1)

