AP Syllabus focus: ‘Particulate models can be used to connect the relative amounts of reactants and products at equilibrium to the value (magnitude) of the equilibrium constant.’
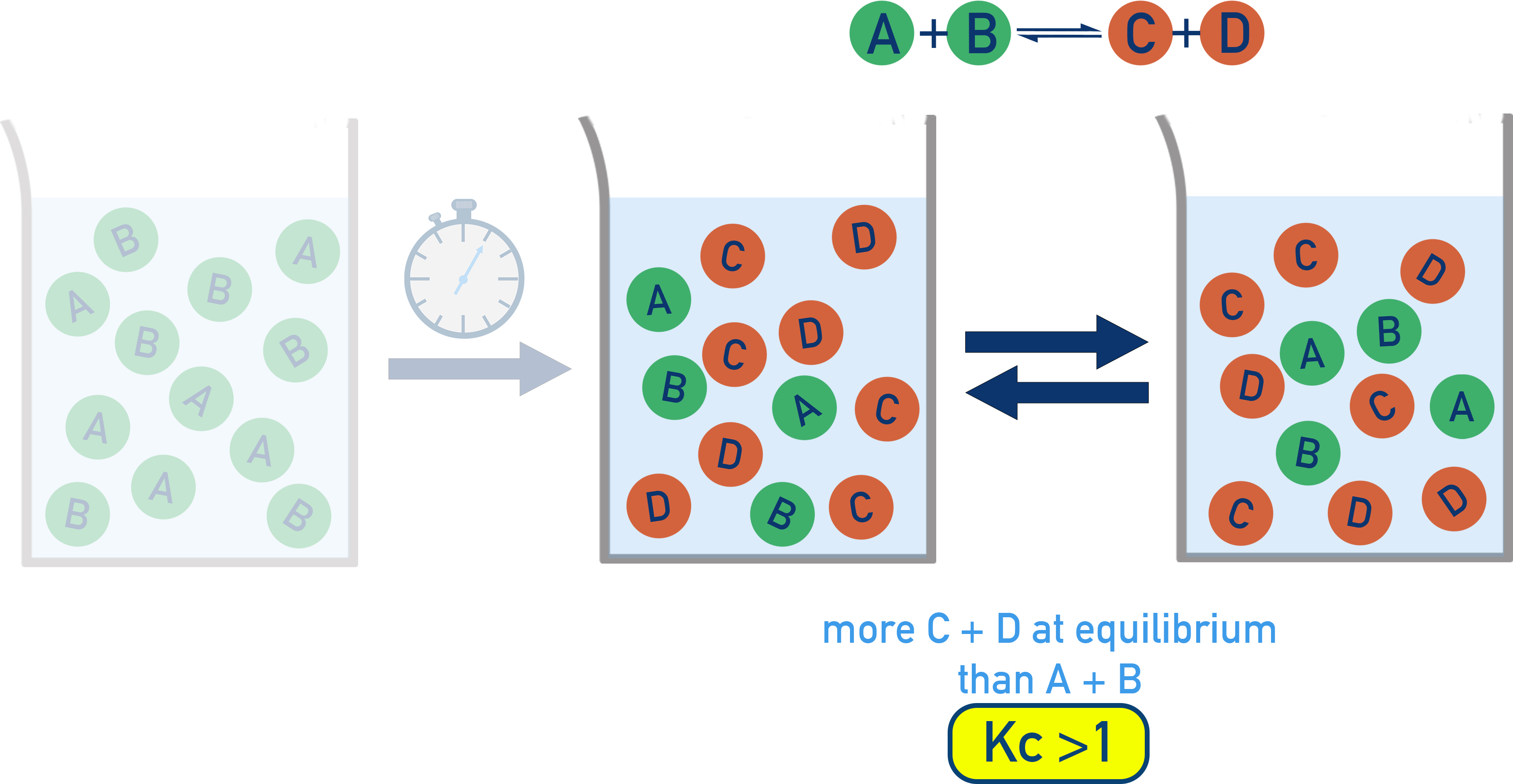
Particle diagrams help you “see” equilibrium composition. By translating particle counts into ratios and then into an equilibrium-constant form, you can infer whether an equilibrium is reactant-favoured or product-favoured.
Connecting particulate pictures to equilibrium composition
At chemical equilibrium, a mixture contains measurable amounts of both reactants and products. A particulate model represents those amounts as numbers of particles (or relative counts) in a fixed volume, letting you compare “how much product” versus “how much reactant” is present once equilibrium is established.
What particle ratios mean (and what they do not)
Particle diagrams can support qualitative and semi-quantitative reasoning:
They show relative amounts of each species at equilibrium (more dots = more of that species).
They can be used to form ratios between species, which is the core idea behind equilibrium-constant expressions.
They do not show reaction rates; equal forward/reverse rates cannot be read from “equal numbers of particles.”
They must be interpreted with the reaction’s balanced coefficients in mind; “twice as many product particles” does not automatically mean “.”
The equilibrium constant as a ratio-based quantity
The equilibrium constant links equilibrium composition to stoichiometry. For a general reaction, the exponents in match the coefficients in the balanced equation, so particle ratios must be processed through that stoichiometric “lens.”
Equilibrium constant (K): A constant (at a given temperature) equal to a ratio of product “amount terms” to reactant “amount terms,” each raised to powers given by the balanced chemical equation.
A particulate model can be treated as a snapshot of relative “amount terms” at equilibrium.
In AP Chemistry contexts, those “amount terms” are typically concentrations; a particle count in a fixed-volume box is proportional to concentration.
= equilibrium constant written using concentrations (unit depends on reaction)
= equilibrium concentration in
= stoichiometric coefficients from the balanced equation
Between different particulate models for the same reaction, the one showing a higher “products-to-reactants” ratio (after applying exponents) corresponds to a larger value.
A particulate (molecular-count) representation for showing an equilibrium mixture dominated by products. The visual connects “more product particles at equilibrium” to a larger equilibrium constant, here labeled . Source
Using particle counts to infer the magnitude of K
Stepwise interpretation (conceptual, not computational)
When given a particulate picture “at equilibrium,” connect it to by reasoning in this order:
Identify the balanced chemical equation (coefficients determine exponents).
Translate the diagram into relative concentrations:
If the volume is the same across diagrams, concentration ratios track particle-count ratios.
If volumes differ, raw counts alone are insufficient (concentration depends on volume).
Build the products over reactants comparison with the correct exponents.
Decide whether is:
Large (product-favoured): equilibrium picture shows mostly products, few reactants.
Small (reactant-favoured): equilibrium picture shows mostly reactants, few products.
Near 1: comparable amounts of reactants and products (after accounting for exponents).
Why stoichiometric exponents matter in particle pictures
Particle counts reflect amounts of species, but reflects an exponent-weighted ratio. This means:
If products have larger coefficients, a “moderate” number of product particles can still correspond to a large because product terms are raised to higher powers.
If reactants have larger coefficients, reactants are exponent-weighted more strongly, so a diagram must be more product-heavy to indicate a large .
A useful way to think: coefficients tell you how many “opportunities” each species has to influence the equilibrium ratio, so your eye should not judge from counts alone without considering those coefficients.
Practical checks and common interpretation pitfalls
Consistency checks for particulate-to-K reasoning
Same container size: Only compare particle counts directly when the diagram implies equal volume.
Species identity: Ensure you are counting the correct species (atoms vs molecules vs ions) as labelled.
Equilibrium snapshot: The diagram must explicitly represent the system at equilibrium; a “before equilibrium” picture does not correspond to .
Relative magnitude only: Particulate models most reliably support comparing whether one scenario has a larger or smaller -like ratio, not determining an exact value.
Frequent mistakes
Assuming “equal numbers of reactant and product particles” means even when coefficients are not 1:1.
Ignoring that is based on a ratio of terms raised to powers, not a simple count ratio.
Comparing two diagrams with different implied volumes as if particle count alone determines concentration.
FAQ
Count the particles as the chemical species shown (molecules, not atoms).
If the diagram shows $A_2$, each “dot-pair” is one particle of $A_2$ for ratio purposes.
Think of the coefficient as amplifying that species’ influence.
A tripled coefficient means the term is raised to the third power, so small changes in that species’ particle ratio can correspond to large changes in the inferred $K$ magnitude.
Yes, if they represent different absolute amounts but the same equilibrium ratio (after applying exponents) at the same temperature.
This is easiest to imagine when the container size or total number of particles differs but relative concentrations satisfy the same expression.
Treat the diagram as proportional, not literal.
Use ratios (e.g., “twice as many”) rather than absolute counts, and then apply stoichiometric exponents to those ratios when reasoning about $K$’s magnitude.
Because “mostly products” is qualitative and can be misleading when coefficients differ.
Also, if reactants appear in small amounts but carry large exponents, they may still strongly affect the ratio that defines $K$, limiting how large $K$ must be.
Practice Questions
(1–3 marks) A particulate diagram for at equilibrium shows many more particles than particles in the same-sized container. What does this imply about the magnitude of ?
States that is greater than 1 / product-favoured (1)
Links reasoning to more than at equilibrium indicating products dominate (1)
Mentions same container size implies counts reflect concentration ratio (1)
(4–6 marks) For , an equilibrium particulate model (fixed volume) shows particles outnumber particles. Explain how the stoichiometric coefficients affect what this suggests about the magnitude of , and why you cannot judge from counts alone.
Writes or describes (1)
Explains coefficients become exponents, so is squared and weighted more strongly (1)
Explains that even if in count, a sizeable can make large, affecting (1)
Concludes you need the exponent-weighted ratio, not raw counts, to infer magnitude (1)
Notes fixed volume allows counts to track relative concentrations (1)
States qualitatively that being high tends to indicate , but not guaranteed without applying the expression (1)

