AP Syllabus focus: ‘When Q is less than K, the reaction proceeds forward overall to form more products; when Q is greater than K, it proceeds in reverse overall to form more reactants.’
Chemical systems rarely start at equilibrium. By comparing the reaction quotient (Q) to the equilibrium constant (K), you can predict the net direction a reversible reaction will proceed to re-establish equilibrium.
Core idea: Q vs K predicts the net change
What Q and K represent
Reaction quotient (Q): A ratio of product amounts to reactant amounts (each raised to stoichiometric coefficients) using the system’s current concentrations or partial pressures.
Q is a “snapshot” of the system right now.
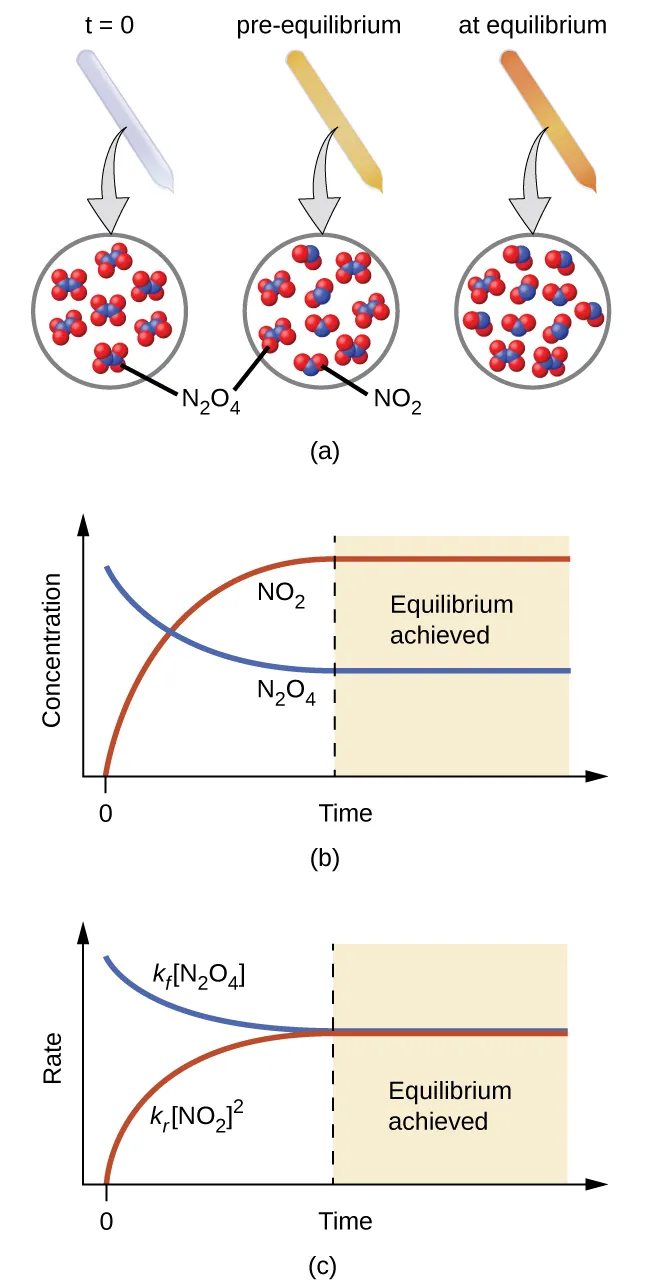
This figure illustrates a reversible system approaching equilibrium using , showing the mixture’s macroscopic change alongside a concentration-versus-time plot. The leveling of the curves represents the point where the composition stops changing (equilibrium), even though microscopic forward and reverse processes continue. This supports the idea that describes the current composition, while equilibrium corresponds to the constant ratio defined by at that temperature. Source
To decide what happens next, compare that snapshot to the target ratio set by K.
Equilibrium constant (K): The value of the same ratio as Q, but specifically when the system is at equilibrium at a given temperature.
K is not a measure of rate or time; it is the equilibrium position expressed as a ratio.
Mathematical form (why Q and K are directly comparable)
= reaction quotient using molar concentrations (unitless for AP purposes)
= molar concentration of species (M)
= stoichiometric coefficients from the balanced equation
For gas-phase systems you may see (using partial pressures); the comparison logic with K is identical as long as you compare like with like (i.e., with , or with ).
Using Q vs K to decide shift direction
The three possible comparisons
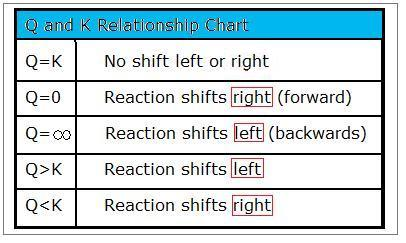
This chart summarizes the three possible relationships between the reaction quotient and the equilibrium constant: , , and . It pairs each comparison with the corresponding net reaction direction needed to re-establish equilibrium (shift toward products or reactants). Source
If : the mixture has too many reactants (or too few products) relative to equilibrium.
Net change: reaction proceeds forward overall to form more products until Q increases to K.
If : the mixture has too many products (or too few reactants) relative to equilibrium.
Net change: reaction proceeds in reverse overall to form more reactants until Q decreases to K.
If : the system is already at equilibrium.
Net change: no net shift (forward and reverse still occur, but cancel macroscopically).
These statements are exactly the syllabus requirement: Q less than K → forward; Q greater than K → reverse.
What “shifts” means (and what it does not mean)
A predicted “shift” is a statement about net reaction progress, not about stopping one direction.
When shifting:
Reactant concentrations/partial pressures decrease in a net forward shift and increase in a net reverse shift.
Product concentrations/partial pressures increase in a net forward shift and decrease in a net reverse shift.
The system stops shifting when the ratio returns to Q = K.
Quick checks and common pitfalls
Check you used the correct K: use with concentrations and with partial pressures.
Use a balanced equation: exponents come from stoichiometric coefficients; the direction decision can be wrong if the reaction is not properly balanced.
Interpret inequality carefully: “ smaller” means “ratio is smaller,” not “all concentrations are smaller.”
Zero/very small terms: if the numerator is (effectively) zero, then Q is very small, so you should expect a forward net reaction to make products.
FAQ
Because direction depends on the reaction’s required ratio (with exponents), not raw counts. Stoichiometric powers can make a small concentration change strongly affect $Q$.
There are essentially no products in the ratio compared with reactants. The system must form products overall, so it proceeds forwards until $Q$ increases to $K$.
Negative: no, because concentrations/partial pressures are non-negative. Undefined can occur if a required denominator term is zero; conceptually $Q \to \infty$, predicting a reverse net direction.
Not directly. $Q$ vs $K$ predicts direction, not rate. Reaction rate depends on kinetics (activation energy, mechanism), not on the equilibrium comparison alone.
Not from those values alone. You must compare like quantities: $Q_c$ with $K_c$ or $Q_p$ with $K_p$. Mixing forms prevents a valid inequality comparison.
Practice Questions
(2 marks) For at a fixed temperature, . A reaction mixture has . Predict the net direction of reaction and state what happens to the amount of .
so reaction proceeds forwards (1)
Amount of increases (1)
(5 marks) For , at a fixed temperature. A mixture has , , .
(a) Write an expression for .
(b) Determine whether the system will proceed forwards or backwards overall.
(1)
Substitution attempt shown (allow ECF): (1)
(1)
Compare with : (1)
Proceeds forwards overall to form more (1)

