AP Syllabus focus: ‘Because K and Q have the same mathematical form, any valid algebraic manipulation applied to K can also be applied to Q.’
Chemical equilibrium problems often require rearranging expressions rather than computing from scratch. Because and share the same structure, the same algebraic moves work for both.
Core idea: same form, same algebra
What it means to “share the same algebra”
Both the reaction quotient and the equilibrium constant are built from the same kind of ratio: products over reactants, with exponents from stoichiometric coefficients. The only difference is when you evaluate them (any time vs. at equilibrium).
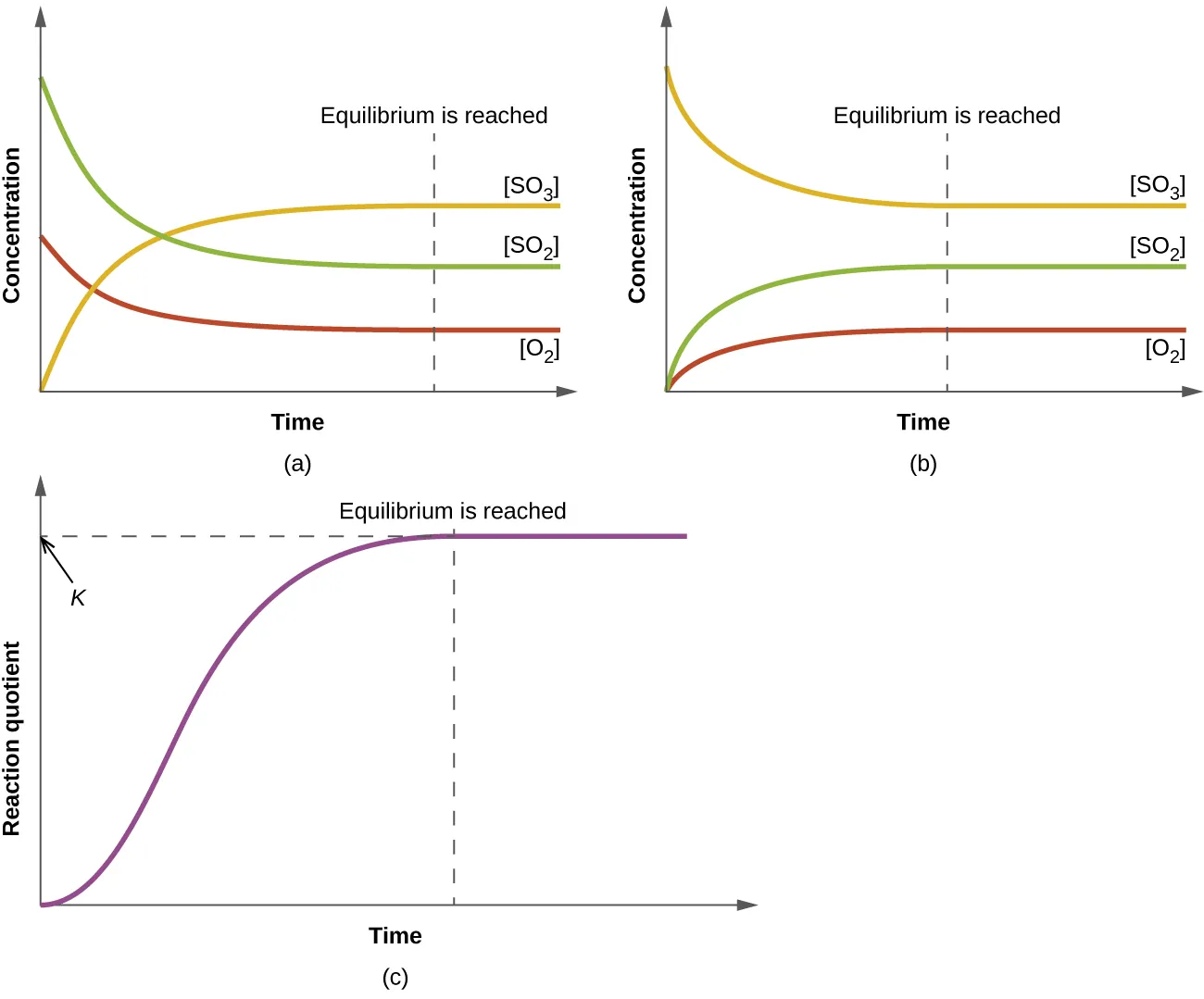
Time-evolution graphs for a reacting system approaching equilibrium, including a plot where the reaction quotient increases and then levels off at a constant value. The plateau corresponds to equilibrium, where stops changing and equals for that temperature. Source
Reaction quotient (Q): A ratio formed from the current amounts (often concentrations or partial pressures) of products to reactants, each raised to their stoichiometric coefficients.
A key consequence is that if you transform a chemical equation in a way that is algebraically valid, the corresponding ratio transforms in the same way.
Equilibrium constant (K): The value of the reaction quotient when the system is at equilibrium (so the ratio has a constant value at a given temperature).
This “same algebra” principle lets you manipulate to compare it to , and it also lets you compute a new for a transformed reaction using the original (or at equilibrium) without rebuilding everything.
Mathematical structure you’re allowed to manipulate
Generic reaction form
For a balanced reaction written as:
the corresponding ratio (whether it is called or depends on conditions) follows the same pattern.
= Stoichiometric coefficients (unitless)
= The measured “amount term” used for that system (commonly concentration or partial pressure in AP Chemistry)
The important algebraic takeaway is not the measurement choice, but that exponents and multiplication/division rules apply identically to both and .
Practical algebraic manipulations (apply identically to Q and K)
Inverting the reaction
If you reverse the chemical equation:
Numerator and denominator swap
The ratio is inverted
So, a valid statement is:
If becomes , then becomes for the reversed reaction using the same mixture state.
Scaling coefficients
If you multiply every coefficient in the reaction by a factor:
All exponents scale by that factor
The whole ratio is raised to that factor
So, a valid statement is:
If becomes , then becomes for the scaled reaction using the same mixture state.
Adding reactions
When reactions are added to make an overall reaction:
Their ratios multiply (because exponents add when like terms combine)
So, a valid statement is:
If overall , then overall QKQKKQ$ expression for that same written reaction.
FAQ
Because adding reactions corresponds to adding stoichiometric changes, which adds exponents for shared species in the overall mass-action form.
Algebraically, multiplying ratios combines powers consistently with exponent rules.
Yes. The algebraic structure (products over reactants with stoichiometric exponents) is the same.
What changes is the “term for species” (activity instead of $[,]$ or $P$), not the exponent/multiplication/inversion rules.
If the chemical equation being manipulated is not chemically meaningful (e.g., adding unrelated reactions without forming a defined net reaction), the resulting quotient may not correspond to any real process.
Also, mixing different “term types” (e.g., some species as partial pressures, others as concentrations) breaks consistency.
Taking $\ln$ (or $\log$) turns multiplication into addition and powers into coefficients.
So if $Q_{\text{new}} = Q^n$, then $\ln Q_{\text{new}} = n\ln Q$, mirroring the same transformation you would apply to $K$.
Using concentrations can make the raw expression look like it has units (e.g., $M$ powers), but thermodynamically $K$ is defined using activities and is dimensionless.
The apparent units come from the concentration approximation and chosen standard state, not from different algebra for $Q$ versus $K$.
Practice Questions
Q1 (1–3 marks) A reaction is written as with reaction quotient . The equation is rewritten as . State the new reaction quotient in terms of .
(1 mark)
Q2 (4–6 marks) You are given two reactions with reaction quotients and at the same moment:
has quotient
has quotient
The overall reaction is formed: . (a) Write the reaction quotient for in terms of and . (b) If the overall reaction is instead written as , write its reaction quotient in terms of and .
(a) States (2 marks: 1 for multiplication idea, 1 for correct expression)
(b) States (2 marks: 1 for recognising squaring when coefficients double, 1 for correct expression)

