AP Syllabus focus: ‘Breaking and forming bonds changes chemical potential energy, which can change particle kinetic energy and appear as a temperature change.’
Chemical reactions often feel hot or cold because energy is redistributed between chemical potential energy stored in bonds and the kinetic energy of particles. That kinetic energy change shows up as a measurable temperature change.
The energy “accounting” behind temperature change
In a reaction, particles collide, old bonds are disrupted, and new bonds are created. Those steps involve changes in stored energy.
Chemical potential energy: Energy stored in the arrangement of atoms and the interparticle attractions (especially chemical bonds) within the system.
A system’s chemical potential energy can be converted to (or from) particle motion. If particle motion increases, the temperature rises; if particle motion decreases, the temperature falls.
Temperature reflects average kinetic energy
Temperature is not “heat” stored in a substance; it is linked to how fast particles move, on average.
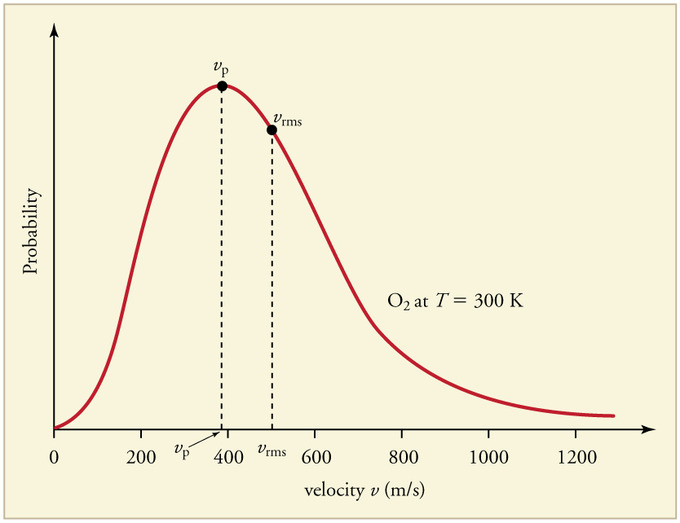
A Maxwell–Boltzmann distribution plot showing how molecular speeds are distributed at a given temperature. The peak marks the most probable speed, while the curve’s width and long high-speed tail emphasize that particles do not all move at the same speed even at one temperature. As temperature increases, the distribution shifts toward higher speeds and broadens, consistent with higher average kinetic energy. Source
Temperature: A measure proportional to the average kinetic energy of the particles in a substance.
This connection is why AP Chemistry treats a temperature change as evidence that energy has moved between stored (potential) and motion (kinetic) forms.
= temperature (K)
= average kinetic energy of particles (J per particle, conceptually)
Bond breaking vs. bond forming: where energy goes
Reactions involve two broad energy contributions that compete.
Breaking bonds requires energy input
To separate bonded atoms, the system must overcome attractive forces.
That energy must come from somewhere (often from particle kinetic energy and collisions, or from the surroundings).
If more energy is required to break existing bonds than is released when new bonds form, particle kinetic energy tends to decrease, so the mixture cools.
Forming bonds releases energy
When new attractions form, the system moves to a lower chemical potential energy state.
The “lost” potential energy is transferred into other forms, commonly:
increased kinetic energy of nearby particles (more vigorous motion)
energy passed outward to surrounding particles through collisions
If bond formation releases more energy than bond breaking consumes, particle kinetic energy tends to increase, so the mixture warms.
How bond-energy changes become a thermometer reading
A thermometer responds to collisions at its surface. Those collisions become more energetic when the reacting mixture’s particles have higher average kinetic energy.
Pathway from bonds to temperature
During reaction progress
Energy is absorbed locally to disrupt bonds and rearrange electrons.
Energy is released locally as new bonds and stable arrangements form.
Redistribution
Energy differences spread through the mixture by collisions.
The balance of these changes determines whether average kinetic energy rises or falls.
Macroscopic observation
Higher average kinetic energy → faster collisions with the thermometer → higher measured temperature.
Lower average kinetic energy → less energetic collisions → lower measured temperature.
Key idea to state clearly on AP responses
A temperature increase does not mean “heat is a product” in a literal substance sense; it means the reaction pathway results in net conversion of chemical potential energy into kinetic energy (and possibly energy transferred outward).
A temperature decrease means the reaction pathway results in net conversion of kinetic energy into chemical potential energy (bond rearrangements that store more energy).
Common conceptual pitfalls (and how to avoid them)
Mistaking temperature for total energy: A system can have high total energy but low temperature if much energy is stored as chemical potential energy rather than kinetic energy.
Ignoring the surroundings: The temperature change you observe depends on energy exchange during and after bond rearrangement; what you measure is the kinetic-energy outcome in the reacting mixture at that time.
Overgeneralising “breaking releases energy”: In chemistry, breaking bonds requires energy, while forming bonds releases energy; the temperature change depends on which effect dominates overall.
FAQ
Both can contribute. Bond rearrangements change chemical potential energy, but changes in attractions between particles (e.g., solvation around ions) can also shift potential energy and therefore kinetic energy.
Energy released into kinetic energy can be spread across many particles, and some energy can be transferred away to the container and air. The observed temperature rise depends on how much energy remains in the reacting mixture as kinetic energy.
Even if the overall process releases energy, early steps may require energy to reach a transition state. That initial energy can be drawn from particle kinetic energy, with later bond formation releasing more energy overall.
Yes. Early bond-breaking-heavy steps can temporarily lower kinetic energy, followed by later bond-formation-heavy steps that raise it, especially if energy redistribution to the surroundings is happening simultaneously.
It means the system moves to a lower (or higher) stored-energy arrangement, and the difference appears as changes in particle motion and collisions. Energy is not destroyed; it is redistributed among available forms.
Practice Questions
(2 marks) Explain, in terms of bonds and particle motion, why a reaction mixture’s temperature can increase during a chemical reaction.
States that forming bonds releases energy / lowers chemical potential energy (1)
Links released energy to increased average kinetic energy of particles, giving higher temperature (1)
(5 marks) A reaction carried out in a beaker causes the solution temperature to drop. Using ideas about bond breaking and bond forming, explain how this temperature decrease can occur. Your answer must refer to chemical potential energy and kinetic energy, and how the thermometer detects the change.
States that breaking bonds requires energy input (1)
States that, overall, more energy is absorbed in bond breaking than is released in bond forming (net energy stored in chemical potential energy) (1)
Explains that this net absorption can come from the reacting mixture’s kinetic energy, decreasing average kinetic energy (1)
Links decreased average kinetic energy to a lower temperature (1)
Explains thermometer detects reduced collision energy/frequency at its surface leading to a lower reading (1)

