AP Syllabus focus: ‘Energy changes can be described as endothermic or exothermic for heating/cooling, phase changes, and chemical reactions.’
Classifying a process as endothermic or exothermic is about tracking whether energy is absorbed by the substance of interest or released from it. This labeling applies consistently across physical changes and chemical reactions.
Endothermic vs. Exothermic: the classification rule
A process is classified by the direction of energy transfer between the material undergoing change and everything else around it (the surroundings).
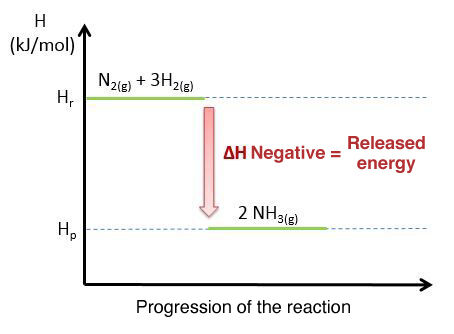
Enthalpy (H) diagram for an exothermic reaction showing products at lower enthalpy than reactants, so . The downward arrow represents energy released from the reacting system to the surroundings. Source
Endothermic process: a process in which energy is transferred into the substance undergoing the change (energy is absorbed).
In practice, endothermic processes often require a continuous energy input (for example, from a hot plate, flame, or ambient thermal energy) to keep the change occurring.
Exothermic process: a process in which energy is transferred out of the substance undergoing the change (energy is released).
When a process is exothermic, energy leaving the changing substance can warm nearby materials (container, solution, air, skin), even if the substance itself is not directly “hot” at every moment.
Heating and cooling (physical temperature changes)
When no phase change or reaction occurs, heating/cooling primarily changes the particles’ average kinetic energy.
Heating (typically endothermic)
Heating a solid, liquid, or gas (with no phase change) is endothermic because energy must be absorbed to raise particle kinetic energy.
The substance gains energy; nearby heat source provides the transfer.
Cooling (typically exothermic)
Cooling a solid, liquid, or gas (with no phase change) is exothermic because energy leaves the substance as particle kinetic energy decreases.
The substance releases energy to its surroundings (for example, to colder air or a cold surface).
Phase changes (physical state changes)
During a phase change, the key energy effect is usually a change in interparticle attractions (potential energy) rather than kinetic energy.
Endothermic phase changes (energy absorbed to separate particles)
Melting (solid → liquid): energy is absorbed to partially overcome attractions holding particles in fixed positions.
Vaporization/boiling (liquid → gas): energy is absorbed to separate particles much more completely.
Sublimation (solid → gas): energy is absorbed to go directly to widely separated particles.
Exothermic phase changes (energy released as particles come together)
Freezing (liquid → solid): energy is released as particles form stronger, more ordered attractions.
Condensation (gas → liquid): energy is released as particles come closer and attractions increase.
Deposition (gas → solid): energy is released as particles form strong attractions in a solid structure.
Chemical reactions (bond changes)
Chemical reactions involve rearranging atoms, which requires breaking some bonds and forming new bonds. The endothermic/exothermic classification depends on the overall balance of energy required versus energy released.
Endothermic reactions (net energy absorbed)
A reaction is endothermic when the energy needed to break bonds (and/or disrupt strong interactions) is greater than the energy released when new bonds form.
The reacting system must absorb energy overall for products to form.
Exothermic reactions (net energy released)
A reaction is exothermic when forming new bonds releases more energy than is required to break the original bonds.
Energy released can appear as heating of the surroundings, light emission, or warming of the reaction vessel.
Quick classification checklist (what AP expects)
Use these cues to classify processes across the three required categories:
Heating/cooling
Heating a substance (no phase change): endothermic
Cooling a substance (no phase change): exothermic
Phase changes
Toward more separated particles (solid→liquid→gas): endothermic
Toward closer, more ordered particles (gas→liquid→solid): exothermic
Chemical reactions
Net bond formation “wins” (more energy released): exothermic
Net bond breaking “wins” (more energy absorbed): endothermic
FAQ
Yes. Many real processes have multiple stages with different energy directions (e.g., initial bond breaking then bond forming). The classification refers to the net energy change overall.
Not necessarily. If the surroundings are much warmer than the reacting mixture, energy may still flow into the system even if the container does not feel cold, or other effects (like evaporation) can mask sensation.
It depends on what you include as the system boundary. If you redefine the system to include the heat source, the direction of energy transfer across the boundary can change.
Activation energy is the minimum energy needed to start a reaction pathway. A reaction can be exothermic overall yet still require an initial energy input to begin.
Often, but not guaranteed. The correct classification always depends on the net balance of energy absorbed in breaking interactions versus released in forming new, more stable interactions.
Practice Questions
(2 marks) State whether each process is endothermic or exothermic. a) Liquid water freezing to ice. b) A metal spoon cooling from to .
a) Exothermic (1)
b) Exothermic (1)
(5 marks) For each process, classify it as endothermic or exothermic and give a brief reason based on particle-level energy ideas. a) Ethanol boiling. b) Nitrogen gas condensing to liquid nitrogen. c) Combustion of methane, .
a) Endothermic (1); energy absorbed to overcome intermolecular attractions/separate particles (1)
b) Exothermic (1); energy released as particles come closer and attractions increase (1)
c) Exothermic (1); forming product bonds releases more energy than is required to break reactant bonds (1)

