AP Syllabus focus: ‘Reaction rates increase with temperature because a larger fraction of collisions can reach the transition state.’
Temperature changes reaction rate by shifting how many molecular collisions have enough energy to form products. Using the transition-state idea, higher temperature means more particles can surmount the energy barrier and react per unit time.
Transition-state idea: why an energy barrier matters
A reacting system must pass through a highest-energy arrangement of atoms before products can form. The key rate question is not “Do particles collide?” but “How many collisions can reach that high-energy configuration?”
Transition state: a short-lived, high-energy arrangement of atoms at the top of the energy barrier along the reaction pathway.
Even in a well-mixed solution or gas, most collisions do not lead to products because the colliding particles separate before reaching the transition state. As temperature rises, a larger fraction of collisions has enough energy to climb the barrier, so the number of successful (reactive) collisions per second increases.
Activation energy as the threshold to reach the transition state
To reach the transition state, colliding particles must supply at least the required energy barrier for the forward reaction.
Activation energy (): the minimum energy required for reactants to reach the transition state and proceed to products.
This “threshold” framing explains the strong temperature sensitivity of rate: small increases in temperature can cause a noticeable increase in the fraction of particles that meet or exceed .
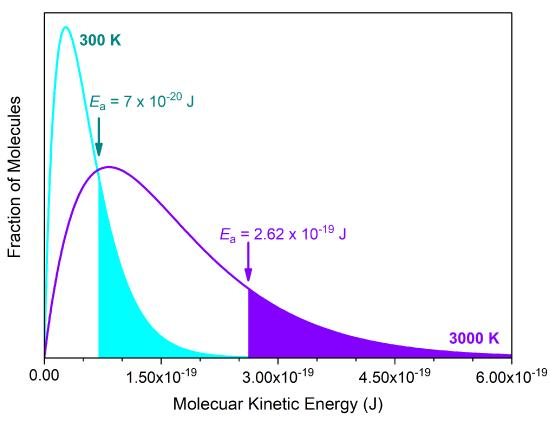
Maxwell–Boltzmann energy distributions at two temperatures with an activation-energy threshold () marked. The higher-temperature curve has a larger high-energy tail, so the shaded area beyond (molecules capable of reacting upon collision) increases, explaining the strong temperature dependence of rate. Source
What increasing temperature does at the particle level
Increasing temperature increases the average kinetic energy of particles and broadens the spread of kinetic energies in the sample. As a result:
Particles move faster, so collisions occur more frequently.
More importantly, a larger fraction of collisions occur with enough energy to reach the transition state.
The second point dominates the observed effect in most chemical reactions. Rate often rises rapidly with temperature because the fraction of sufficiently energetic collisions is very sensitive to temperature.
= kinetic energy available during a collision (J per collision, conceptually)
= activation energy barrier to reach the transition state (J per collision, conceptually)
Only collisions meeting this condition can reasonably proceed to products; raising temperature increases the proportion of collisions satisfying it.
“Larger fraction” means more successful collisions per unit time
At any moment, particles in a sample have a distribution of kinetic energies.
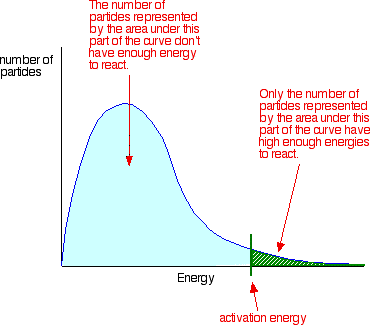
Maxwell–Boltzmann distribution with the activation energy marked as a cutoff on the energy axis. The area to the right of the cutoff represents the fraction of particles with enough kinetic energy to react upon collision, connecting the distribution picture to the idea of “successful collisions per unit time.” Source
At lower temperature, relatively few particles populate the high-energy tail of that distribution, so relatively few collisions can reach the transition state. At higher temperature, that high-energy tail becomes more populated, so:
More collisions can access the transition state
More reacting systems cross the barrier per second
The observed macroscopic rate (change in concentration per time) increases
This is the transition-state interpretation of the syllabus statement: temperature increases rate because it increases the fraction of collisions capable of reaching the transition state.
What temperature does not change (in this model)
Raising temperature does not change the balanced chemical equation or the reaction stoichiometry. It also does not “force” low-energy collisions to react; instead, it changes the statistics of energies so a greater share of collisions are above the barrier.
In the transition-state picture, temperature primarily changes:
how often the system can form the transition state and not:
the identity of reactants/products
the net amount of reactant needed per product formed
Linking the idea to energy profiles (conceptually)
On a reaction energy profile, the transition state appears at the peak (highest point).
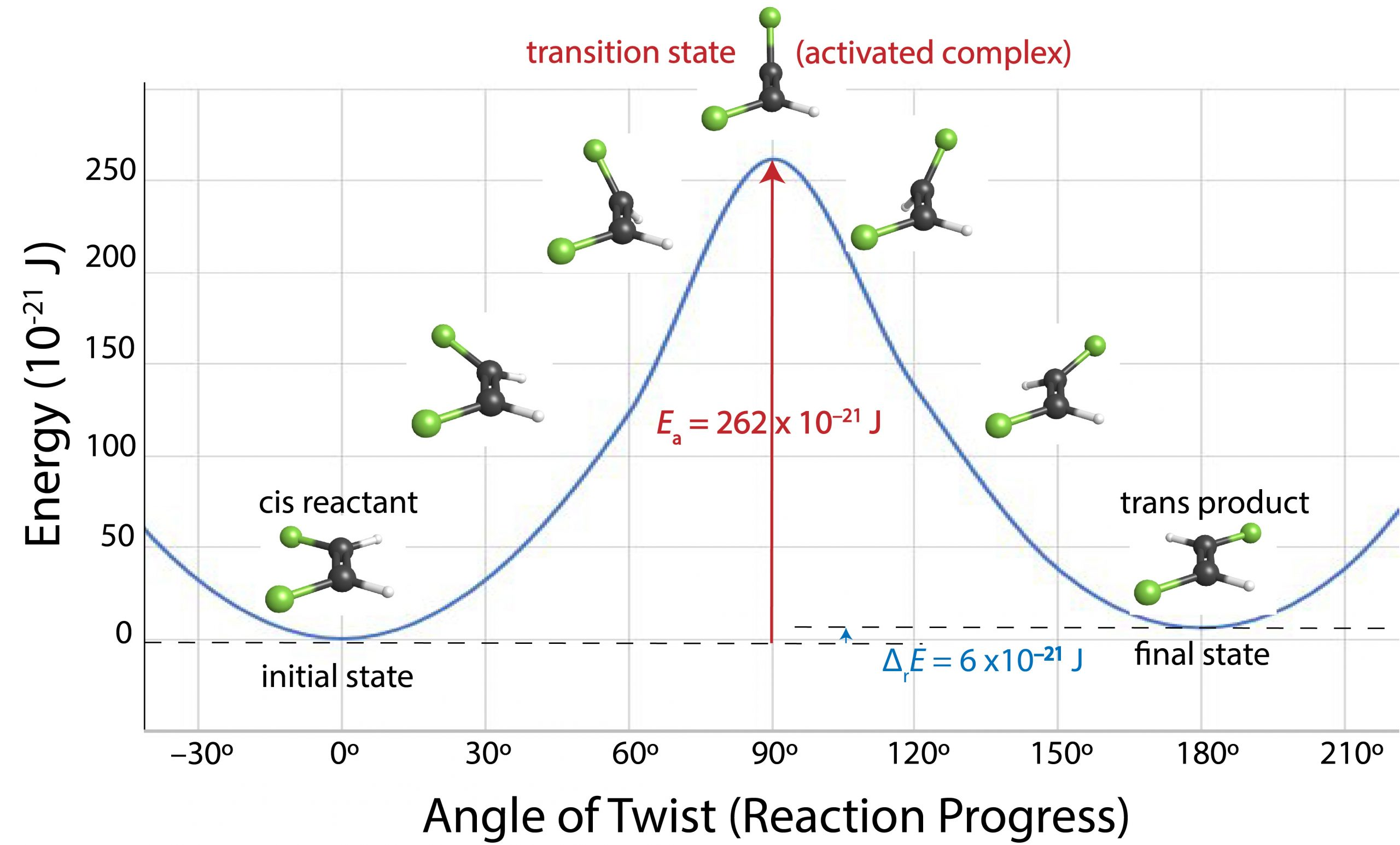
Reaction energy (reaction coordinate) diagram illustrating a single-step pathway with a maximum-energy transition state. The vertical difference between the reactants and the peak corresponds to the forward activation energy , making the “energy barrier” interpretation of temperature effects concrete. Source
The vertical gap from reactants up to that peak corresponds to the forward activation energy. When temperature increases, the peak height does not necessarily change, but more collisions have enough energy to reach that peak. Therefore, more trajectories proceed over the barrier to products, increasing the reaction rate.
FAQ
No. Reaching the transition state means the system gets to the top of the barrier.
From there, it can proceed to products or revert to reactants.
Because the population of particles above $E_a$ lies in the high-energy tail.
That tail’s population is highly temperature-sensitive.
For typical elementary barrier-controlled reactions, yes.
If other constraints dominate (e.g., diffusion limits), the increase may be smaller.
Collision frequency is how often particles hit.
The fraction is the proportion of those hits energetic enough to reach the transition state.
A larger $E_a$ means fewer collisions exceed the threshold at low temperature.
So increasing temperature usually causes a more dramatic fractional increase in successful collisions.
Practice Questions
(2 marks) Using the transition-state idea, explain why increasing temperature increases reaction rate.
Mentions that a larger fraction of collisions has sufficient energy to reach the transition state / overcome . (1)
Links this to more successful (reactive) collisions per unit time, increasing the rate. (1)
(5 marks) A reaction has a significant activation energy barrier. Describe, in terms of collision energies and the transition state, how raising the temperature affects (i) the fraction of collisions that lead to reaction and (ii) the observed rate.
States that particles have a distribution of kinetic energies. (1)
Explains that increasing temperature increases the average kinetic energy (or shifts population to higher energies). (1)
States that a greater fraction of collisions have and can reach the transition state. (2)
Concludes that more successful collisions per unit time increases the observed reaction rate. (1)

