AP Syllabus focus: ‘For a zero‑order reaction, [A] decreases linearly with time: [A]t − [A]0 = −kt.’
Zero-order kinetics describes reactions whose rates do not depend on reactant concentration. This page focuses on how concentration changes over time, how to recognise linear behaviour, and how to interpret the integrated rate law graphically.
Core idea: zero-order concentration–time behaviour
What “zero order” means
Zero-order reaction: A reaction whose rate is independent of the reactant concentration; the rate stays constant as long as the reaction conditions remain the same.
In a zero-order process, the reactant is consumed at a constant rate, so equal amounts of reactant disappear in equal time intervals. This commonly arises when a surface or active site is saturated, so adding more reactant does not increase the number of effective events per second.
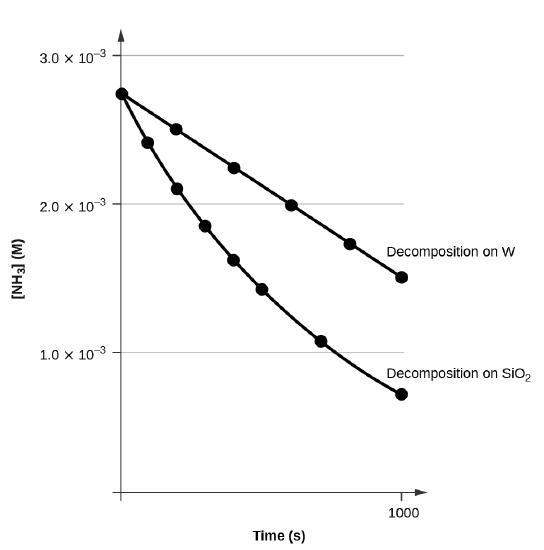
A concentration–time plot compares the decomposition of on two different surfaces. The straight-line trace illustrates zero-order kinetics (constant consumption rate), while the curved trace illustrates non-zero-order behavior under different surface conditions. This kind of comparison highlights why surface saturation can produce an apparent zero-order rate law over a measured interval. Source
The integrated rate law (zero order)
For a single reactant being consumed, the relationship between concentration and time is linear.
= concentration of at time (typically )
= initial concentration of (typically )
= elapsed time (units depend on the experiment, e.g. )
= zero-order rate constant (typically )
This equation encodes the syllabus statement directly: decreases linearly with time for a zero-order reaction.
Linear plots: what to graph and how to read it
The correct linear graph
A hallmark of zero-order kinetics is that plotting versus yields a straight line (assuming the reaction remains zero order over the measured interval).
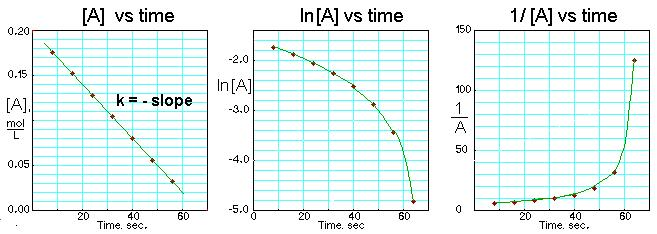
Three standard “order-test” plots for the same dataset are compared side-by-side. For a zero-order reaction, the vs. plot is linear, and the slope magnitude gives the rate constant via . The other transformed plots are non-linear, helping you visually confirm the reaction order. Source
Key features of the vs. line:
Slope is negative for a disappearing reactant (because concentration decreases with time).
-intercept equals at .
The line remains straight only while the reaction truly follows zero-order behaviour (constant rate).
Interpreting the slope without overextending
Because the integrated law is in the form , the slope corresponds to for reactant consumption. Practically, this means:
A steeper downward slope indicates a larger and faster consumption of .
Units of slope are concentration/time, matching the required units of in zero-order kinetics.
Physical and experimental implications
Constant-rate consumption and “run-out” time
Since decreases at a constant rate, the model predicts that will eventually reach zero after a finite time if zero-order conditions persist. In real data:
You should not treat the line as valid once is nearly exhausted or conditions change.
Apparent deviations from linearity at late times often reflect changing conditions (for example, loss of saturation).
Data-quality checks specific to zero-order plots
When assessing whether a dataset is consistent with zero order, focus on:
Whether vs. is approximately linear across the measured interval.
Whether the extrapolated intercept is reasonable (close to the measured initial concentration).
Whether the measured concentration ever becomes “negative” by extrapolation (a sign you have extended the model beyond its physically meaningful domain).
Common interpretation pitfalls
A straight-line decrease in with time supports zero-order behaviour, but only if concentration measurements are reliable and conditions are stable.
If the rate appears constant only over part of the run, report zero-order behaviour only over that interval; many real systems shift order as conditions evolve.
FAQ
Zero order often indicates a limiting “capacity” such as a saturated surface or fixed number of active sites.
If all sites are occupied, increasing $[A]$ does not increase the number of effective events per second, so the rate stays constant.
Use the idea that $[A]$ drops by equal amounts in equal times.
On a $[A]$ vs $t$ straight line, the “run-out” time is where the line would reach $[A]=0$; this estimate is only meaningful while zero-order behaviour holds.
Common causes include loss of saturation, changes in temperature, or depletion that alters the controlling condition.
Once the controlling capacity is no longer the bottleneck, the kinetics may shift away from zero order, producing curvature.
No. Many reactions show decreasing $[A]$ with time; zero order is indicated specifically by an approximately constant slope (linearity) over the relevant interval.
A short, noisy dataset can look linear by chance, so check consistency and experimental uncertainty.
The numerical value of $k$ depends on the chosen time unit.
If you switch from seconds to minutes, $k$ must change so that $kt$ is unchanged; the units become $\mathrm{M,min^{-1}}$ instead of $\mathrm{M,s^{-1}}$.
Practice Questions
(2 marks) A reaction consumes reactant with zero-order kinetics. On which graph would the data form a straight line: vs , vs , or vs ? State one feature of the straight line.
Identifies vs as linear (1)
States a correct feature: intercept or slope (1)
(5 marks) The concentration of is monitored during a reaction. The plot of against time is a straight line with negative gradient. (a) Deduce the order with respect to and justify using the integrated rate law form. (3) (b) State the units of the rate constant for this reaction, with a brief reason. (2)
Deduces zero order (1)
Uses integrated law idea: or (1)
Links linearity of vs (and gradient ) to zero order (1) (b)
States units (or concentration/time consistent with given time unit) (1)
Reason: slope has units concentration/time and slope corresponds to (1)

