AP Syllabus focus: ‘Construct balanced redox equations by writing and combining oxidation and reduction half-reactions to produce an overall balanced equation.’
Balancing redox reactions can look intimidating because both mass and charge must balance. The half-reaction method provides a reliable, stepwise procedure that tracks electrons explicitly and produces a chemically consistent overall equation.
Core idea: split the reaction into two electron processes
In any redox reaction, one species undergoes oxidation (loses electrons) and another undergoes reduction (gains electrons).
The half-reaction method balances each process separately, then recombines them so electrons cancel.
Half-reaction: An equation showing either oxidation or reduction alone, including explicit electrons () to account for charge changes.
A correct final redox equation must conserve atoms and overall charge, which is ensured by matching electrons lost and gained.
= number of electrons transferred (dimensionless count)
Half-reaction method in acidic aqueous solution
Use these steps when the reaction occurs in acidic solution (or when and are reasonable species to include).
1) Write the unbalanced overall (skeleton) equation
Keep correct formulas for reactants and products.
Split into two half-reactions: one oxidation, one reduction.
2) Balance all elements except H and O
In each half-reaction, balance atoms other than H and O by adjusting coefficients.
3) Balance oxygen with water
Add to the side needing oxygen atoms.
Recheck all non-H atoms after adding water.
4) Balance hydrogen with
Add to the side needing hydrogen atoms.
This step is specific to acidic conditions.
5) Balance charge with electrons
Compute total charge on each side of the half-reaction.
Add to the more positive side until charges match.
Interpretation:
Electrons on the product side indicate oxidation (electrons produced).
Electrons on the reactant side indicate reduction (electrons consumed).
6) Equalise electrons and add the half-reactions
Multiply one or both half-reactions by integers so the number of electrons cancels when added.
Add the half-reactions together and cancel identical species on both sides (including , and sometimes or ).
7) Final checks
Atom check: each element has the same count on both sides.
Charge check: total charge matches on both sides.
Coefficient check: use the smallest whole-number coefficients.
Adapting the method to basic aqueous solution
If the redox reaction occurs in basic solution, a common approach is:
First balance as if acidic (using , , and ).
Then eliminate using .
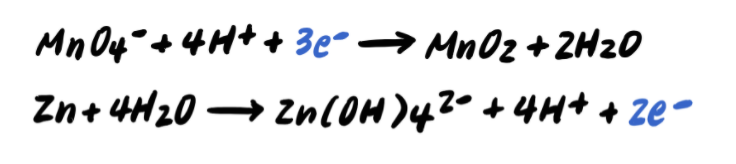
Two half-reactions are shown after adding to both sides, demonstrating the formal neutralization step used to remove in basic media. This diagram supports the idea that you add the same amount of to preserve both mass and charge before simplifying to water and canceling spectators. Source
Converting an acidic-balanced result to basic
Add the same number of to both sides as there are .
Combine on each side where they appear.
Cancel any molecules that appear on both sides after combining.
Recheck atom and charge balance.
Common pitfalls and quick diagnostics
Forgetting to balance charge (not just atoms) in each half-reaction.
Adding or to the wrong side (always add to fix the deficit).
Failing to multiply all coefficients in a half-reaction when scaling to equalise electrons.
Ending with electrons in the final overall equation (they must cancel).
Not reducing coefficients to lowest whole-number terms.
FAQ
Electrons are bookkeeping devices for charge balance within half-reactions. In the full reaction, electrons are transferred internally, so they must cancel.
After balancing charge, $e^-$ on the products side indicates oxidation; $e^-$ on the reactants side indicates reduction.
In aqueous balancing, $H_2O$ can be introduced to balance oxygen because water is the solvent and available as a reactant/product.
Adding $OH^-$ to both sides preserves equality, then neutralises $H^+$ by forming $H_2O$ without changing overall stoichiometry.
Not strictly; half-equations can be set up from reactants/products. Oxidation numbers can help identify what is oxidised/reduced to choose the split correctly.
Practice Questions
(2 marks) In acidic solution, which species is added to balance (i) oxygen atoms and (ii) hydrogen atoms when using the half-equation method?
(i) (1)
(ii) (1)
(6 marks) Balance the following redox equation in acidic solution using half-equations:
Writes correct reduction half-equation with and balances O/H using and : (2)
Writes correct oxidation half-equation: (1)
Multiplies iron half-equation by 5 to equalise electrons (1)
Adds and cancels to obtain: (2)

