AP Syllabus focus: ‘When acids or bases ionize in water, identify conjugate pairs and compare their relative strengths.’
Comparing acid and base strengths in water becomes much easier when you organise species into conjugate acid–base pairs. The key idea is that proton transfer creates linked partners whose strengths are inversely related.
Key idea: conjugate pairs link strengths
Conjugate partners in proton transfer
Conjugate acid–base pair: Two species that differ by exactly one proton (); the acid has one more than its conjugate base.

This diagram summarizes a generic Brønsted–Lowry proton-transfer reaction, . It visually identifies the two conjugate pairs ( and ) created by a single transfer, reinforcing how conjugates differ by exactly one proton. Source
In water, many reactions can be viewed as competition for between bases, or competition to donate between acids. Each proton-transfer step creates two conjugate pairs (one from the acid, one from the base).
What “strength” means in aqueous solution
Acid (or base) strength: A measure of the extent to which an acid donates to water (or a base accepts from water), reflected by the position of the ionisation equilibrium.
A stronger acid ionises more in water (equilibrium lies further toward products) and therefore has a larger dissociation constant and smaller .
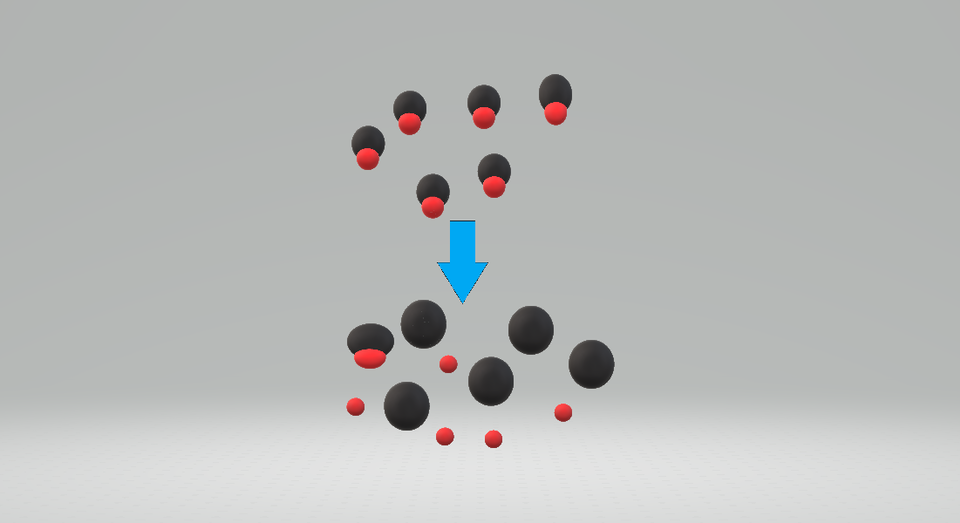
This particle diagram illustrates a strong acid in water as mostly (or essentially completely) dissociated into ions rather than remaining as intact acid molecules. It provides a microscopic interpretation of “ionises more,” which corresponds to larger and smaller in aqueous solution. Source
A stronger base accepts protons more readily (larger , smaller ).
Comparing strengths using conjugate pairs
The inverse-strength rule (most tested idea)
For a conjugate pair in water:
If is a stronger acid, then must be a weaker conjugate base (it has little tendency to accept back).
If is a stronger base, then must be a weaker conjugate acid.
This inverse relationship is the central shortcut: you can often rank base strength by ranking the strengths of their conjugate acids (in the opposite order), and vice versa.
Using equilibrium direction to compare strengths (no numbers required)
For a generic proton-transfer reaction in water:
Identify the two acids present (a reactant acid and a product acid).
Identify the two bases present (a reactant base and a product base).
The equilibrium favours formation of the weaker acid and weaker base.
So, when comparing conjugate pairs:
If products contain the weaker acid/base pair, the forward reaction is more favoured.
If reactants contain the weaker pair, the reverse reaction is more favoured.
Using , , , and with conjugates
= acid dissociation constant (unitless)
= equilibrium concentration of species in
= base dissociation constant (unitless)
= ion-product constant for water (at , )
When and are conjugates in water, their strengths are mathematically linked:
Larger for means smaller for (since ).
On a logarithmic scale: smaller for corresponds to larger for .
Practical workflow for AP questions
Write the species as conjugate pairs (look for “differ by one ”).
Use the inverse-strength rule to convert an acid-strength comparison into a base-strength comparison (or vice versa).
If given values, rank acids by lower = stronger acid, then reverse the order to rank their conjugate bases.
If only reaction direction is provided, decide which side contains the weaker acid/base pair.
Common pitfalls to avoid
Don’t confuse concentration with strength: a dilute strong acid is still strong; strength depends on extent of ionisation, not how much is present.
Don’t compare bases by looking at the base formulas alone; compare their conjugate acids whenever possible.
Remember that species can be amphiprotic; always identify which role (acid or base) it is playing in the specific reaction before assigning its conjugate partner.
FAQ
Very strong acids appear equally strong in water because they transfer $H^+$ essentially completely to form $H_3O^+$.
This makes their conjugate bases appear extremely weak in water, limiting how finely you can rank them experimentally.
Because conjugate pairs satisfy $K_aK_b=K_w$, changing $K_a$ forces an opposite change in $K_b$.
On a log scale, a decrease of 1 unit in $pK_a$ corresponds to a tenfold increase in $K_a$ and thus a tenfold decrease in $K_b$ for the conjugate base.
No. A conjugate pair differs by exactly one $H^+$, so a given acid has a unique conjugate base (and vice versa).
However, a species can have more than one possible conjugate partner if it can lose different protons in different steps.
First decide whether it is acting as an acid or a base in the given reaction context.
Then form the appropriate conjugate pair for that role only, and apply the inverse-strength rule within that specific pairing.
Equilibrium composition measurements can be used to infer whether products or reactants are favoured, indicating the weaker acid/base side.
Spectroscopic or conductivity changes consistent with greater ionisation can also support “stronger acid/base” claims in aqueous solution.
Practice Questions
(1–3 marks) The conjugate acids of , , and have values of 3.2, −7, and −9 respectively. Rank the bases , , and from strongest to weakest.
Uses to rank acids: lower = stronger acid (1)
States conjugate base strength is inverse to conjugate acid strength (1)
Correct base order: (1)
(4–6 marks) Consider the equilibrium: . (a) Identify the two conjugate acid–base pairs.
(b) Explain, using conjugate-pair strength reasoning, why is a stronger base than in water.
(a) Correct pair: (1)
(a) Correct pair: (1)
(b) States equilibrium favours weaker acid/base pair (1)
(b) Identifies product-side acid/base are weaker: weaker acid than and/or weaker base than (1)
(b) Uses inverse-strength logic: weaker conjugate acid implies stronger conjugate base (1)
(b) Concludes stronger base than consistent with equilibrium position (1)

