AP Syllabus focus: ‘Chemical processes typically involve breaking and/or forming chemical bonds; physical processes typically involve changes in intermolecular interactions (e.g., phase changes).’
Chemical and physical changes can look similar in the lab, but AP Chemistry distinguishes them by what happens at the particle level. Track whether intramolecular bonds change or whether only intermolecular attractions change.
Core distinction: what changes at the particle level?
Chemical processes: bonds change
Chemical change is identified by breaking and/or forming chemical bonds within particles (atoms, molecules, or ions). This rearranges atoms into new combinations, producing new substances with different formulas and properties.
Chemical change: A process in which chemical bonds are broken and/or formed, changing the identity (composition) of the substances present.
Because bonds define how atoms are connected, changing bonds changes the set of particles in the system (new molecules/ions form), not just their spacing or motion.
Physical processes: intermolecular forces change
Physical change is identified by changes in intermolecular interactions and particle arrangement, while the identities of the particles remain the same.
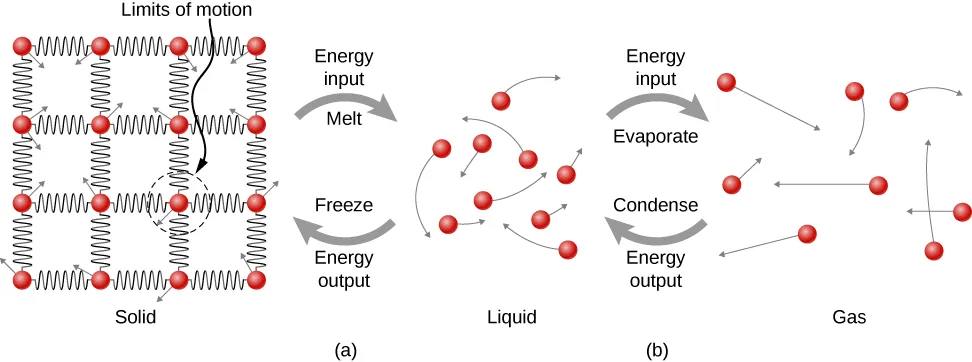
This figure models phase changes with particle-level diagrams: a solid with closely packed particles, a liquid with mobile but still interacting particles, and a gas with widely separated particles. The labeled energy arrows highlight that melting and evaporation require energy input to overcome attractions, while freezing and condensation release energy as attractions become more extensive. Source
Common cases are phase changes (melting, boiling, freezing, condensing) and many mixing processes.
Intermolecular forces (IMFs): Attractions between particles (e.g., dispersion forces, dipole–dipole attractions, hydrogen bonding) that influence physical properties like boiling point and viscosity.
A key idea is that physical changes typically adjust how close particles are and how freely they move, without altering the covalent bonds inside molecules or the ionic structure of ions.
How to recognise each type in representations
Using particulate diagrams
Chemical change diagrams show particles that are connected differently after the change (new groupings/structures), indicating bond rearrangement.
Physical change diagrams show the same particles before and after, but with different spacing/ordering (solid-like lattice vs liquid disorder vs gas separation).
Using formulas and names
If the process produces a substance with a new formula (not just a new state symbol), it points to chemical change.
If only state symbols change, such as , it points to physical change because the substance’s composition is unchanged.
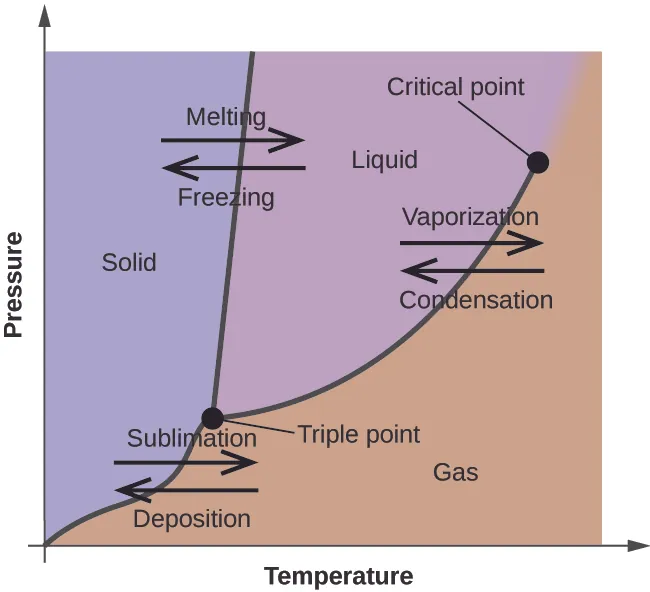
This phase diagram divides conditions of temperature and pressure into regions where a substance is solid, liquid, or gas, with boundary lines representing equilibrium between phases. It visually reinforces that moving from to is a change in physical state driven by conditions—not a change in chemical identity. Source
Energy perspective (why heat isn’t enough to decide)
Both types of change can involve heat transfer, so temperature change alone does not prove chemical change.
Chemical processes involve breaking/forming bonds, often with larger energy changes per mole because covalent/ionic bonds are relatively strong.
Physical processes involve overcoming or strengthening IMFs; these are usually smaller energy changes, but can still be noticeable (for example, boiling requires significant energy input).
Typical examples aligned to the distinction
Physical (IMFs change; composition unchanged)
Melting ice: water molecules remain , but hydrogen-bonding patterns and molecular motion change.
Boiling ethanol: ethanol molecules remain intact; IMFs are overcome as molecules separate into the gas phase.
Condensation: IMFs form more extensively as particles come closer.
Chemical (bonds change; new substances form)
Decomposition or synthesis reactions (new molecules with new bonding).
Any process where atoms are rearranged into different bonded groupings (even if phase also changes).
Common pitfalls
“Gas formed” can occur in a physical process (evaporation) or a chemical reaction (new gaseous product). Decide by whether the gas is the same substance as before.
Color/heat changes can accompany both; rely on particle-level bond changes rather than observations alone.
A process may involve both physical and chemical steps; for AP classification here, focus on whether the central change is bond rearrangement or IMF rearrangement.
FAQ
Look for changes in composition using analytical evidence (e.g., new substances detectable by spectroscopy or chromatography) rather than appearance alone.
A physical change keeps molecular identity constant.
IMFs arise from partial/temporary charge attractions between particles, while chemical bonds involve electron sharing or full charge attraction in an ionic lattice.
This difference typically makes IMFs much lower in energy.
For most molecular substances, phase changes rearrange spacing and IMFs only.
Exceptions are uncommon in introductory AP contexts and usually involve changes to extended structures rather than typical liquids and gases.
Even though each IMF interaction is weak, a mole of liquid contains an enormous number of interacting particles.
Separating them into a gas requires overcoming many attractions overall.
Higher external pressure favours particles staying closer together, supporting condensed phases where IMFs can act more effectively.
Lower pressure makes it easier for particles to separate into the gas phase.
Practice Questions
(2 marks) Explain, in particle terms, the difference between a chemical process and a physical process.
1 mark: Chemical process involves breaking and/or forming chemical bonds (new substances/particles).
1 mark: Physical process involves changes in intermolecular interactions/arrangement without changing composition.
(5 marks) A student heats a liquid sample and observes bubbling. Describe what evidence would distinguish boiling from a chemical reaction that produces a gas, using bonding and intermolecular ideas.
1 mark: Boiling is a physical change where particles are the same substance before/after.
1 mark: Boiling involves overcoming intermolecular forces, not breaking intramolecular bonds.
1 mark: Chemical gas formation involves bond breaking/forming to make a new gaseous substance.
1 mark: Reference to composition/formula staying the same (physical) vs changing (chemical).
1 mark: Mention that heat/bubbles alone are not definitive; must identify whether identity changes.

