AP Syllabus focus: ‘Physical changes alter properties (including phase or mixing) without changing a substance’s composition.’
Matter can undergo many observable changes in the lab. In AP Chemistry, the key distinction is whether a process changes chemical composition (identity) or only changes physical properties like phase, particle arrangement, or mixing.
Core idea: properties vs composition
Chemical composition (what a substance is)
Composition: The kinds of particles present (atoms, ions, molecules) and their ratios, which determine a substance’s chemical identity and formula.
A sample’s composition stays the same if the same particles are present in the same chemical ratios, even if the sample’s appearance changes. Many measured properties (color intensity, density, conductivity, temperature, volume) can change without any change in composition.
Physical properties (what a substance looks like or how it behaves)
Physical properties describe a substance without changing what it is. Common property changes include:
Phase (solid, liquid, gas)
Temperature (warming/cooling)
Shape and size (cutting, grinding)
Distribution in space (mixing, separating mixtures physically)
Physical change: composition unchanged
Physical change: A process that changes physical properties (including phase or mixing) while leaving chemical composition unchanged.
A physical change rearranges particles or changes their spacing/energy, but does not create new chemical species.
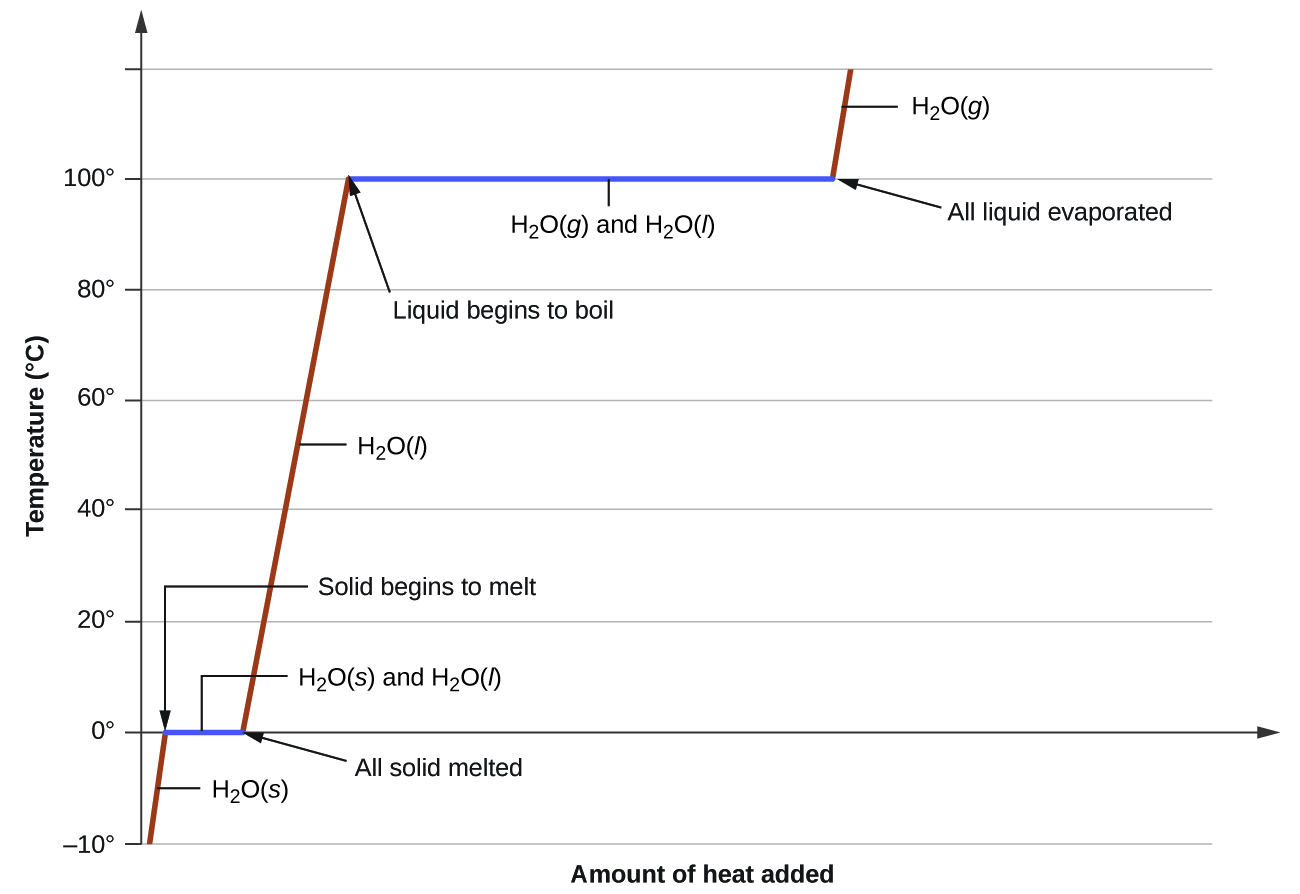
Heating curve for water illustrating how temperature changes as heat is added, with flat plateaus at phase transitions. During these plateaus, added energy goes into overcoming intermolecular forces (latent heat) rather than increasing temperature, so the substance’s chemical identity remains unchanged. Source
At the particle level:
In a phase change, the particles are the same before and after; only intermolecular attractions and particle spacing/ordering change.
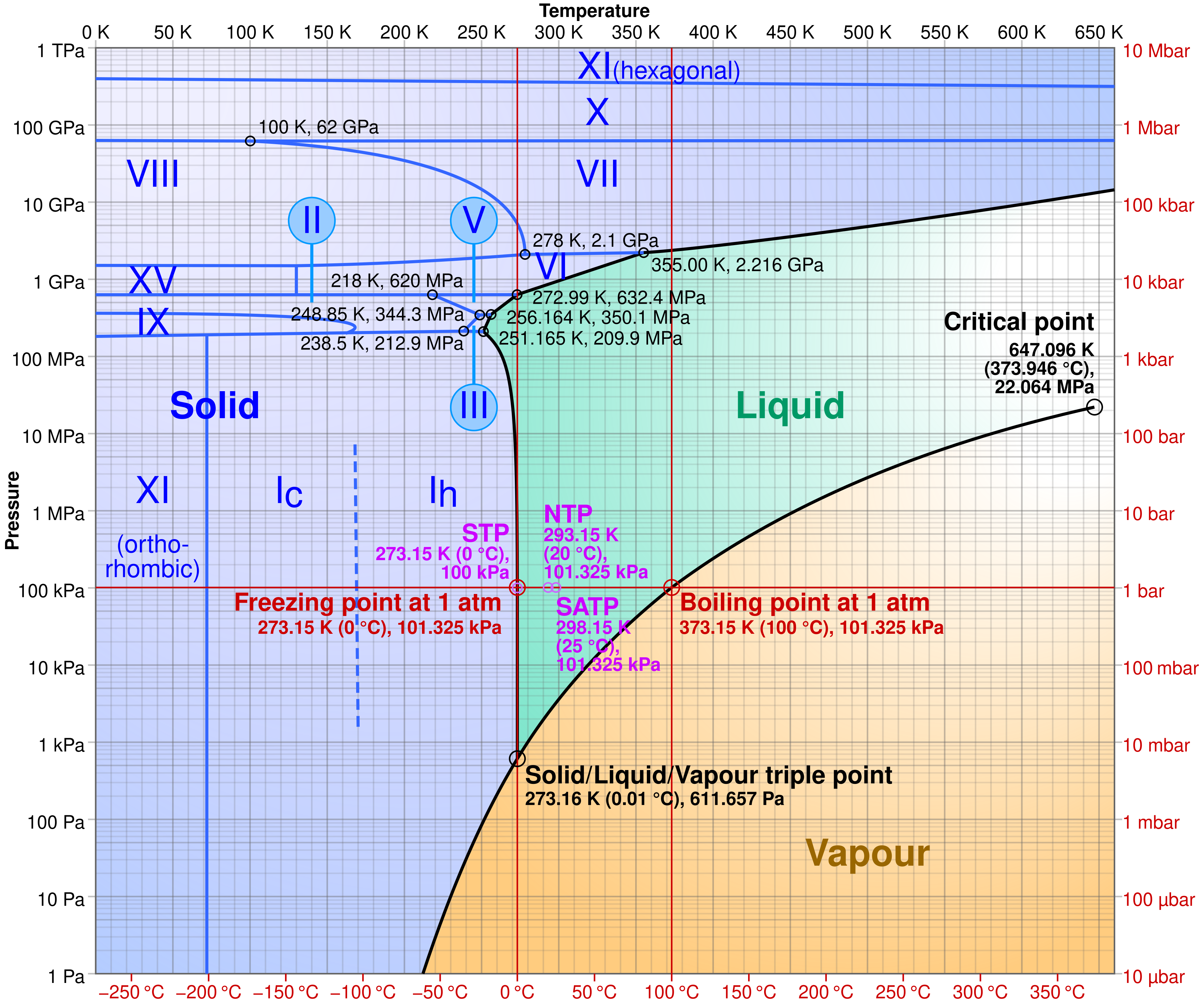
Phase diagram of water showing which phase (solid, liquid, or gas) is thermodynamically stable at different temperatures and pressures. The boundary curves represent equilibrium between phases, and crossing a boundary corresponds to a phase change without changing chemical composition (still H2O). Source
In mixing, different substances become interspersed, but each substance retains its own identity.
Common physical changes you should recognise in AP Chemistry include:
Melting/freezing (e.g., ice to liquid water)
Boiling/condensing
Sublimation/deposition
Mixing two substances without reaction (e.g., combining sand and iron filings)
Grinding/crushing a solid (smaller pieces, same composition)
Important nuance: a dramatic appearance change can still be physical. For example, crushing a crystal changes surface area and light scattering, but not composition.
Chemical change (contrast): composition changed
A chemical change occurs when composition changes—meaning at least some particles are transformed into different particles with different bonding and/or different ratios of atoms. In AP Chemistry terms, chemical change implies:
New substances are formed (new identities)
Chemical bonds are broken and/or formed
Reactant formulas differ from product formulas
Do not rely on “easy” rules like “irreversible = chemical”: some physical changes are hard to reverse under typical conditions, and some chemical changes can be driven backward.
How to decide quickly (AP-style reasoning)
Use identity-first thinking
Ask: after the process, do you still have the same substances you started with?
If yes, it’s a physical change (composition unchanged).
If no, it’s a chemical change (composition changed).
Look at what would be written in formulas
Physical changes can often be represented by changing only the state label while keeping the formula the same (for example, water solid to water liquid). Chemical changes require different formulas for products than for reactants.
Mixing vs reacting
“Mixing” is explicitly part of the physical-change idea in the syllabus statement:
Physical mixing: substances coexist; composition of each substance remains unchanged.
Chemical reaction during mixing: composition changes (new species appear), so it is not merely a physical change.
When evaluating a description, separate:
What is being done (heating, cooling, stirring, grinding) from
What is being produced (same substances vs new substances)
FAQ
Compare an intensive identifier before and after (e.g., melting point after resolidifying).
Use instrumental methods (IR, mass spectrometry) to confirm the same molecular signature.
Energy can go into overcoming intermolecular attractions rather than breaking intramolecular bonds.
The particles remain the same; only their arrangement/spacing changes.
Many properties depend on particle spacing and mobility (e.g., viscosity, conductivity).
Large property changes do not imply new chemical species.
Yes—changing surface area (grinding) or phase can change collision frequency.
This affects rate, not composition at the moment of the physical change.
A pure substance has a fixed particle identity and ratio; a mixture contains multiple substances together.
Mixing changes the sample’s overall makeup, but not each substance’s chemical identity.
Practice Questions
(2 marks) State whether melting solid iodine to liquid iodine is a physical or chemical change. Justify your answer using the idea of composition.
1 mark: Identifies it as a physical change.
1 mark: Justifies that the composition/identity remains iodine (same particles), only the phase changes.
(6 marks) For each process below, state whether it is a physical change or a chemical change, and give a brief particle-level explanation focused on whether composition changes. (a) Grinding crystalline sugar into a fine powder. (b) Mixing iron filings with sand. (c) Passing an electric current through molten sodium chloride and collecting two different substances at the electrodes.
(a) 1 mark physical; 1 mark explanation: same sugar molecules/composition, only particle size/surface area changes.
(b) 1 mark physical; 1 mark explanation: iron and sand particles are just intermingled; no new substances/composition unchanged.
(c) 1 mark chemical; 1 mark explanation: new substances form (different particles than NaCl), so composition changes.

