AP Syllabus focus: 'Spectrophotometers are set to the wavelength of maximum absorbance (optimum wavelength) for the species to maximize sensitivity in Beer–Lambert measurements.'
Choosing the correct wavelength and operating a spectrophotometer properly are essential for accurate concentration measurements. This page focuses on selecting and the practical steps that connect instrument settings to sensitivity.
Why is chosen
A spectrophotometer measures how much light a sample absorbs at a selected wavelength.
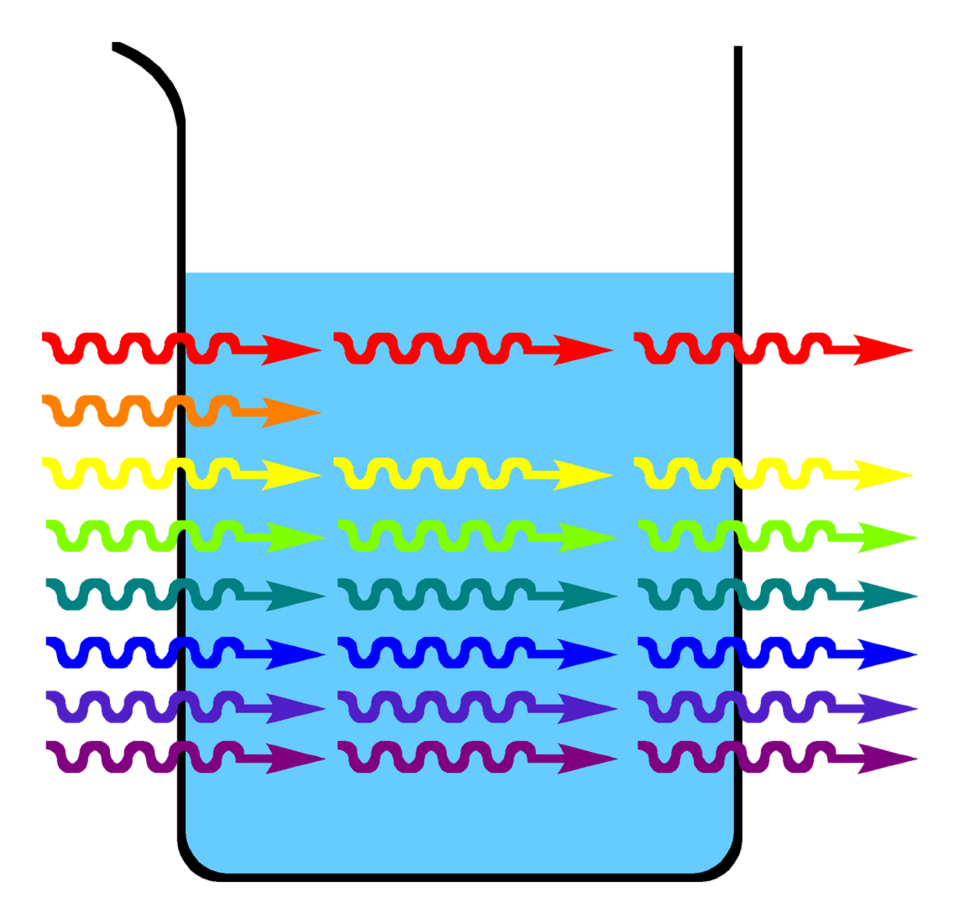
Conceptual diagram showing that a blue solution absorbs orange light (complementary color) more strongly, so the transmitted light differs in spectral composition from the incident light. This illustrates the core idea of wavelength-dependent absorption that underlies choosing an optimal measurement wavelength. Source
For a given analyte, absorbance varies with wavelength, producing an absorption spectrum with a peak.
: the wavelength at which a species shows its maximum absorbance under the measurement conditions.
Setting the instrument to makes the method most sensitive to concentration changes because absorbance responds most strongly there.
= absorbance (unitless)
= molar absorptivity at the chosen wavelength (L·mol·cm)
= path length through the sample (cm)
= concentration of the absorbing species (mol·L)
At , is largest, so the slope of an absorbance vs. concentration relationship is steepest.
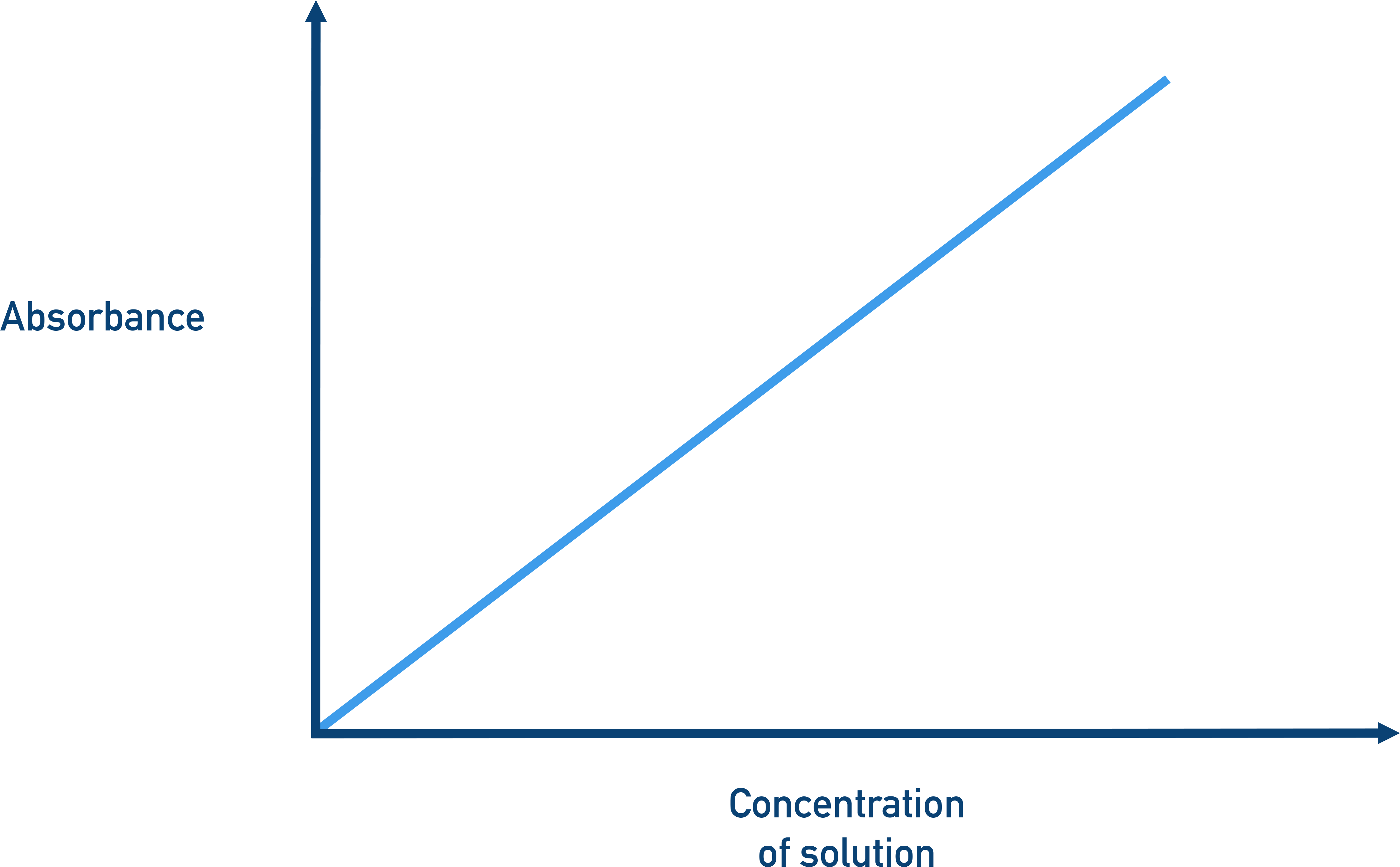
A linear absorbance-versus-concentration graph illustrating Beer–Lambert behavior under conditions where and path length are constant. The straight-line relationship emphasizes why spectrophotometry can use a calibration curve to determine unknown concentrations from measured absorbance. Source
This “maximizes sensitivity” because a small change in produces a larger, more measurable change in .
Practical benefits beyond sensitivity
Improved precision: near the top of a peak, small wavelength-setting drift often changes less than it would on a steep side-slope.
Better signal-to-background: if other components absorb less at , selectivity can improve (while still targeting the analyte’s strongest response).
How to find the optimum wavelength
In many labs, is provided. If it is not, it is determined by scanning.
Determining (conceptual workflow)
Prepare a solution containing the absorbing species (at a concentration that gives a measurable, not saturated, absorbance).
Run a wavelength scan over a relevant range (often visible if the solution is coloured).
Identify the wavelength of the highest absorbance peak; use that as for subsequent measurements.
Using a spectrophotometer correctly (measurement workflow)
Good technique ensures the wavelength choice actually translates into reliable absorbance data.
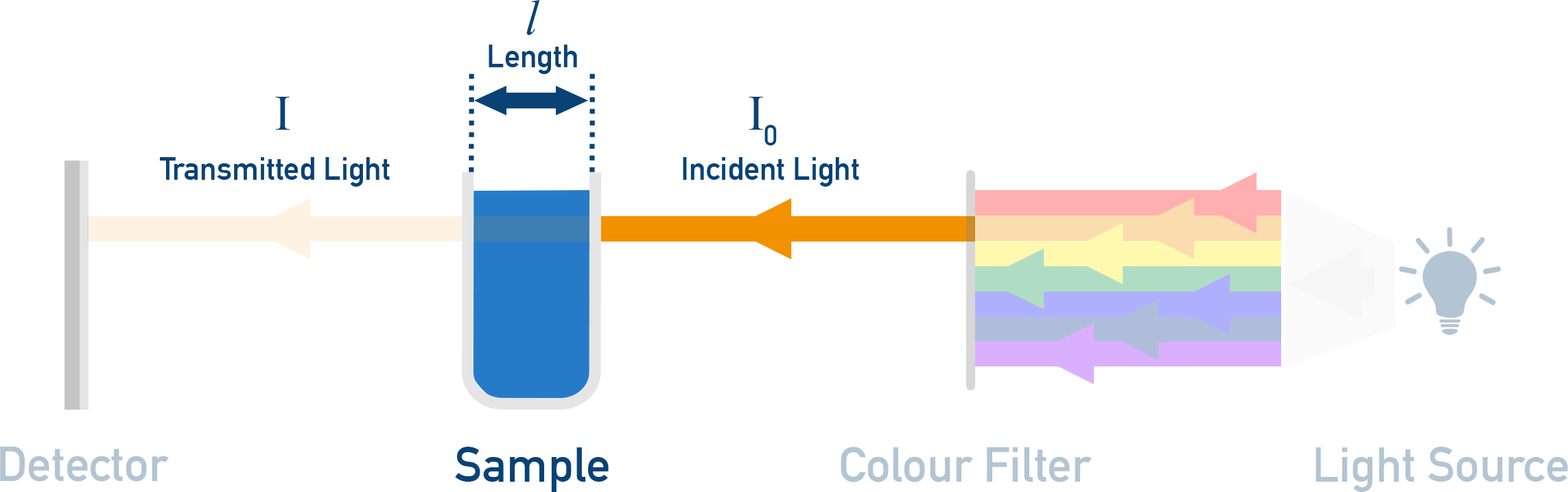
Schematic of a simple spectrophotometer/colourimeter optical path: polychromatic light is filtered to a selected wavelength, passes through the sample cuvette (path length), and the detector compares transmitted intensity to the incident beam. This diagram helps visualize why consistent cuvette orientation and a fixed path length are essential for reliable absorbance measurements. Source
Blank: a reference solution containing the solvent (and any reagents except the absorbing analyte) used to set the instrument’s baseline absorbance.
Core operational steps
Select the correct mode (typically absorbance, not % transmittance).
Set the wavelength to the analyte’s (the “optimum wavelength”).
Fill a clean cuvette with the blank, wipe the clear faces, insert with consistent orientation, and zero the instrument.
Measure samples in matching cuvettes (same type and path length) with consistent handling and orientation.
Record absorbance values only after the reading stabilises.
Handling details that matter for data quality
Keep the cuvette’s optical faces clean and dry; hold by the frosted/ridged sides.
Ensure no bubbles are in the light path and the sample is well-mixed.
Use the same cuvette type (glass vs plastic) across blank/standards/unknowns to avoid wavelength-dependent differences.
FAQ
If the absorption band is broad, any wavelength near the top can work.
A practical approach is to choose the centre of the plateau where small wavelength shifts change absorbance least, improving robustness.
Yes. Changes in chemical form or environment can shift $\lambda_{\max}$.
Re-scan under the exact conditions used for measurement (same solvent, pH, reagent concentrations) to ensure the selected wavelength matches the absorbing species present.
Very high absorbance means very little light reaches the detector, which can reduce measurement reliability.
In practice, you adjust concentration (e.g., dilution) so readings fall in a comfortable instrumental range while still using $\lambda_{\max}$.
A spectrophotometer selects a narrow wavelength (monochromator), allowing precise choice of $\lambda_{\max}$.
A colorimeter uses broader filters; you pick the filter that best overlaps the analyte’s absorption peak rather than a single exact wavelength.
Material choice depends on wavelength transmission.
Plastic cuvettes may absorb in parts of the UV; glass has limits too. For UV work, quartz is often required. Always match cuvette material to the wavelength region being used.
Practice Questions
(2 marks) Explain why a spectrophotometer is set to when using Beer–Lambert measurements.
States that absorbance is greatest at / is largest at (1)
Links this to maximum sensitivity: a small change in concentration gives a larger change in absorbance / steeper gradient (1)
(5 marks) A student must measure the concentration of a coloured ion using a spectrophotometer. Describe how they would select an appropriate wavelength and then obtain absorbance readings for their solutions.
Describes obtaining/using an absorption spectrum to identify (1)
States to set the spectrophotometer to (1)
Describes using a blank to zero/baseline the instrument (1)
Mentions consistent cuvette use (same path length/type and consistent orientation) (1)
Mentions an operational detail that supports valid readings (e.g., wipe cuvette faces, avoid bubbles, mix, allow reading to stabilise) (1)

