AP Syllabus focus: ‘Hydrogen bonding occurs when H bonded to N, O, or F is attracted to the negative end of a dipole on N, O, or F in another molecule or another part of the same molecule.’
Hydrogen bonding is a particularly strong, directional intermolecular attraction that appears in specific polar molecules. Understanding exactly when it forms and why it is unusually strong helps you predict structure and behaviour in molecular substances.
What hydrogen bonding is (and is not)
Hydrogen bonding is best viewed as a strengthened, highly directional dipole–dipole interaction that occurs only under specific bonding and lone-pair conditions.
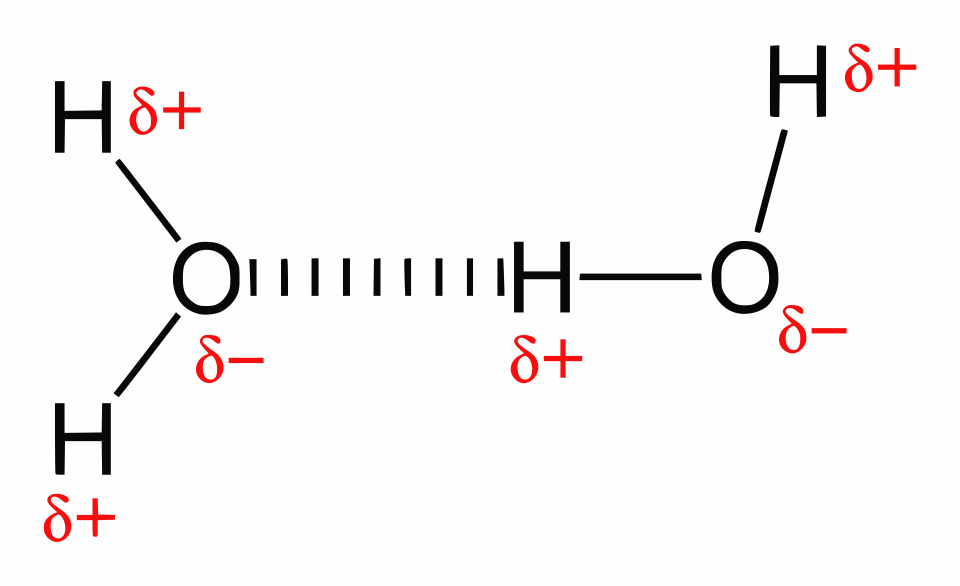
This diagram shows a hydrogen-bonding network in liquid water, with dotted lines indicating hydrogen bonds between molecules. The partial charges ( on H and on O) highlight why the attraction is fundamentally electrostatic, while the consistent O–H···O alignment illustrates hydrogen bonding’s strong directionality. Source
Hydrogen bond: An attractive interaction between a hydrogen atom covalently bonded to N, O, or F and a lone pair on N, O, or F in a nearby molecule or a different part of the same molecule.
Hydrogen bonding is not a covalent bond; the molecules remain distinct chemical species, held together by an intermolecular attraction (or held in a particular shape by an intramolecular attraction).
Required features: donor and acceptor
A hydrogen bond forms only when both of the following are present:
A hydrogen-bond donor: a polar bond H–N, H–O, or H–F, where H carries a substantial partial positive charge ().
A hydrogen-bond acceptor: an atom N, O, or F with at least one lone pair that provides a region of high electron density (the negative end of a dipole, ).
Hydrogen-bond donor/acceptor: A donor is an H attached to N/O/F; an acceptor is a nearby N/O/F lone pair that can attract that H.
A molecule may be a donor, an acceptor, both, or neither depending on its structure (for example, an –OH group can donate and accept).
When hydrogen bonding occurs
Hydrogen bonding occurs in two main contexts that match the syllabus wording.
Intermolecular hydrogen bonding (between molecules)
This occurs when the donor group on one molecule aligns toward an acceptor atom on another molecule:
H–N, H–O, or H–F provides the hydrogen
A nearby N, O, or F provides the lone-pair region
The interaction is strongest when the atoms approach closely and align well (directional attraction)
Intermolecular hydrogen bonding commonly leads to clusters or extended networks when many donors and acceptors are available.
Intramolecular hydrogen bonding (within the same molecule)
Hydrogen bonding can also occur “in another part of the same molecule” when a single molecule contains:
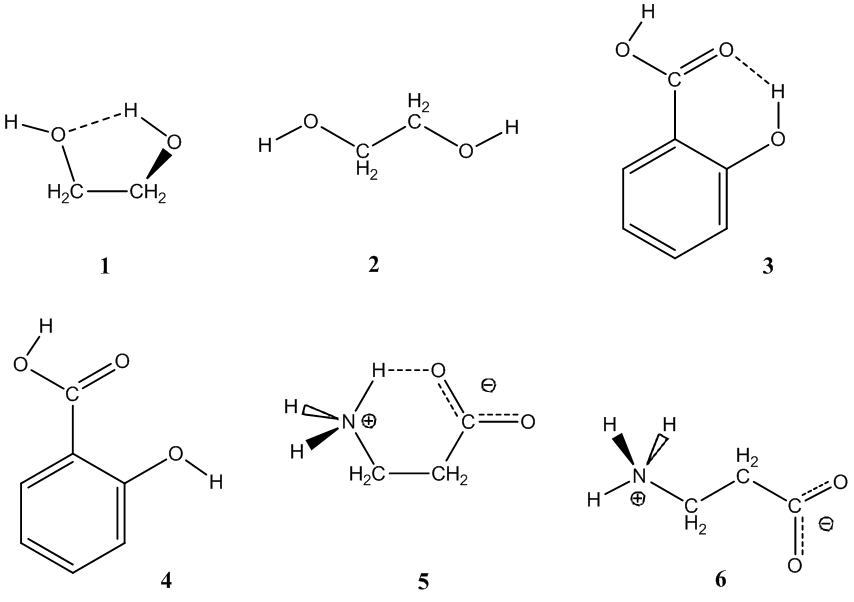
This figure illustrates examples of intramolecular hydrogen bonding, where a donor H on an O–H or N–H group bends back toward a nearby lone-pair-bearing atom within the same molecule (shown with dashed lines). By contrasting conformations that do and do not form the internal H-bond, it makes clear how molecular geometry controls whether intramolecular hydrogen bonding can occur. Source
A donor group (H–N/O/F)
An acceptor atom (N/O/F with lone pairs)
A geometry that allows the donor hydrogen to approach the acceptor closely (often creating a stabilising folded shape)
Intramolecular hydrogen bonding competes with intermolecular hydrogen bonding; if a molecule internally “satisfies” its donor/acceptor sites, it may hydrogen-bond less with neighbouring molecules.
Why hydrogen bonding is strong
Hydrogen bonding is stronger than typical dipole–dipole attractions because of the unusual charge distribution and close approach enabled by N, O, and F.
Large partial charges from high electronegativity
N, O, and F are highly electronegative, so bonds to hydrogen are extremely polar:
Electron density is pulled toward N/O/F, leaving H significantly
The acceptor atom has a concentrated region associated with its lone pairs
The strong attraction increases the magnitude of the Coulombic interaction compared with many other polar molecules.
Close approach due to small atomic size
Hydrogen is very small, so the donor hydrogen can approach an acceptor lone pair closely. Shorter distance between opposite charges increases attraction, making hydrogen bonding unusually strong for a neutral-molecule interaction.
Directionality (orientation matters)
Hydrogen bonds are highly directional, meaning strength depends strongly on how the molecules are oriented:
Strongest when the donor bond points toward the acceptor lone pair (an “aligned” approach)
Weaker when the geometry forces a bent or misaligned interaction
This directionality helps explain why hydrogen bonding can impose specific shapes (especially when intramolecular) and why some structures form extended networks more readily than others.
Why only N, O, and F (in AP Chem context)
Hydrogen bonding in AP Chemistry is restricted to H bonded to N, O, or F because these atoms combine:
Very high electronegativity (strong bond polarity)
Small size (high charge density and effective close contact)
Lone pairs capable of acting as strong acceptor sites
Other atoms can form weaker analogous attractions, but they are not treated as “hydrogen bonding” in the standard AP definition and are typically much less significant.
Recognising hydrogen bonding in structures
When deciding if a substance can hydrogen-bond, check for the structural requirements rather than relying on molecular formula alone:
Look for –OH, –NH, or HF units (donors)
Look for O, N, or F with lone pairs (acceptors)
Verify that the acceptor is available (for example, an atom lacking lone pairs cannot accept)
In mixtures, hydrogen bonding can also occur between different species if one provides the donor and the other provides an acceptor site, provided N/O/F are involved as specified.
FAQ
No. Hydrogen bonding is a local interaction: it depends on having an H–N/O/F donor and an N/O/F lone-pair acceptor.
A molecule can have hydrogen-bonding groups yet have a small net dipole if its bond dipoles cancel.
Typically no. A positively charged nitrogen generally lacks an available lone pair, so it cannot accept a hydrogen bond.
It may still act as a donor if it has N–H bonds.
The acceptor’s electron density is concentrated in lone-pair regions, not spread uniformly around the atom.
This makes the interaction strongest when the donor H points towards the lone-pair region, so orientation changes strength noticeably.
It can stabilise a folded conformation by “holding” two parts of the same molecule together.
This is most effective when the donor and acceptor can reach each other without severe strain and the alignment is favourable.
No; strength varies with:
Donor/acceptor identity (O vs N vs F)
Distance between H and the acceptor atom
Alignment (more linear tends to be stronger)
Local environment (nearby groups can enhance or reduce electron density at the acceptor)
Practice Questions
Question 2 (4–6 marks) A molecule contains both an –OH group and a nitrogen atom with a lone pair elsewhere in the same molecule. (a) Describe how this molecule could form a hydrogen bond within a single molecule. (b) State two structural conditions that would make this intramolecular hydrogen bond more likely or stronger. (c) Explain briefly why hydrogen bonding is stronger than ordinary dipole–dipole interactions.
Mentions H on O–H (donor) attracted to lone pair on N (acceptor) within the same molecule (1)
Links to attraction between H and region on N (1) (b) Any two, one mark each (2)
Donor and acceptor positioned close enough to approach
Geometry allows favourable alignment/directionality (proper orientation)
Minimal steric hindrance allowing close contact
Conformation that brings –OH and N into proximity (e.g., ring/chain flexibility enabling contact) (c)
Explains larger partial charges due to high electronegativity of N/O/F and strong bond polarity (1)
Explains stronger attraction from short distance/close approach and/or high charge density and directionality (1)
Question 1 (1–3 marks) Methanol, , forms hydrogen bonds but chloromethane, , does not. Explain why.
Identifies that hydrogen bonding requires H covalently bonded to N, O, or F (1)
States that methanol has an O–H bond so H is sufficiently and oxygen has lone pairs to act as an acceptor (1)
States that chloromethane has no H bonded to N/O/F (and Cl is not used for hydrogen bonding in AP definition), so it cannot hydrogen-bond (1)

